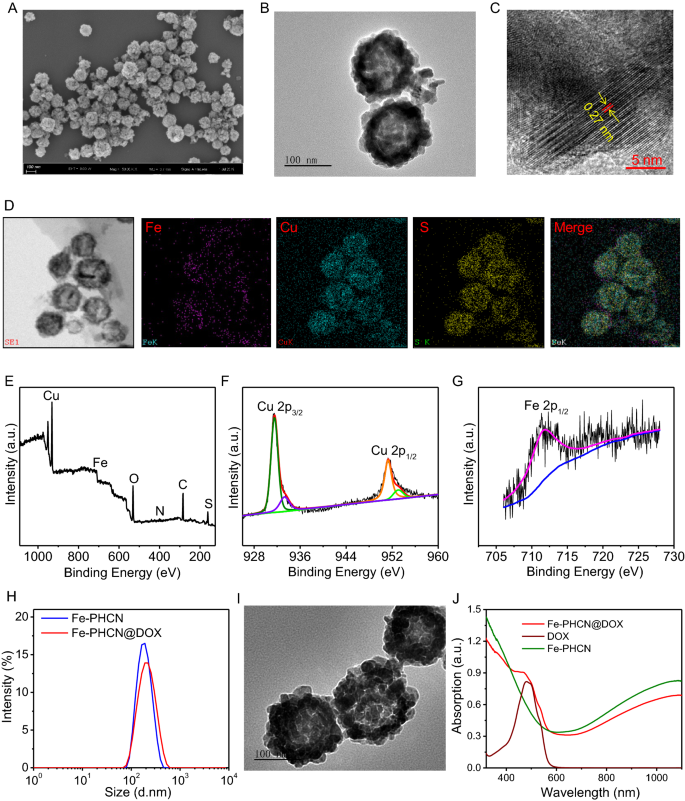
Preparation and characterization of Fe-PHCN@DOX nano-nuclear-reactors
Right here, the Fe-PHCN nano-nuclear-reactors had been facilely ready as described within the Experiment Part by introduction of Fe3+ to PHCN nanomaterials (Fig. 1). Fe-PHCN nano-nuclear-reactors had been used for efficient loading DOX to assemble the multifunctional Fe-PHCN@DOX nano-nuclear-reactors. Consultant scanning electron microscopy (SEM) confirmed that the Fe-PHCN nano-nuclear-reactors exhibited wonderful dispersion and monodisperse spherical form (Fig. 2A; Extra file 1: Fig. S1). Transmission electron microscopy (TEM) pictures confirmed that Fe-PHCN nano-nuclear-reactors possessed hole traits and uniform dimension roughly 130 nm (Fig. 2B; Extra file 1: Fig. S2). The nano-nuclear-reactors with a thickness shell of ≈ 20 nm exhibited an apparent hole construction. The hydrodynamic diameter of Fe-PHCN nano-nuclear-reactors decided by dynamic gentle scattering (DLS) was roughly 191 nm (Extra file 1: Fig. S3), which was barely greater than TEM common diameter as a result of hydration impact. The lattice spacing (d-spacing) of the nano-nuclear-reactors was about 0.27 nm (Fig. 2C). The composition evaluation of the nano-nuclear-reactors by way of TEM elemental mapping with Fe, Cu, and S components was noticed, suggesting Fe efficiently doped to PHCN nanomaterials (Fig. 2D). X-ray photoelectron spectroscopy (XPS) and energy-dispersive X-ray spectroscopy (EDX) revealed that Fe, Cu, S, O, C, N had been introduced within the pattern, additional demonstrating the profitable preparation of Fe-PHCN nano-nuclear-reactors (Fig. 2E; Extra file 1: Fig. S4). Within the XPS information (Fig. 2F), the peaks positioned at 951.3/953.1 and 931.5/933.3 eV respectively correspond to Cu 2p1/2 and Cu 2p3/2 ranges, confirming the coexistence of Cu+ and Cu2+. In the meantime, the attribute peaks of 711.5 eV accord with the Fe 2p1/2 (Fig. 2G), suggesting the existence of Fe3+ in nano-nuclear-reactors [46]. The zeta potential dropped from − 19.3 mV all the way down to − 13.05 mV after PEGylation (Extra file 1: Fig. S5), demonstrating the profitable modification of PEG. To additional affirm the modification of PEG, we measured the FT-IR spectra of Fe-HCN and Fe-PHCN. FT-IR spectra clearly demonstrated that the particularly attribute peaks for –C–O–C– had been clearly enhanced after PEG modification (Extra file 1: Fig. S6), indicating the profitable modification of PEG on nano-nuclear-reactors. The UV–vis absorption spectra confirmed the Fe-PHCN options possessed sturdy absorbance within the NIR I and II area (Extra file 1: Fig. S7).
Characterization of the nano-nuclear-reactors. SEM (A) and TEM (B) picture of Fe-PHCN. The dimensions bar represents 100 nm. C HRTEM pictures of Fe-PHCN. The dimensions bar represents 5 nm. D EDS elemental mapping of Fe-PHCN. E XPS characterization of Fe-PHCN. XPS high-resolution scans of Cu (F) and Fe (G) in Fe-PHCN. H Dimension of Fe-PHCN and Fe-PHCN@DOX by DLS measurement. I TEM picture of Fe-PHCN@DOX. The dimensions bar represents 100 nm. J UV–vis spectrum of Fe-PHCN, DOX, and Fe-PHCN@DOX
DOX and the as-prepared Fe-PHCN had been combined to arrange Fe-PHCN@DOX nano-nuclear-reactors after stirring and centrifugation. Due to their uniquely hole porous construction, the nano-nuclear-reactors could possibly be used as a drug nanocarrier. Small DOX molecules (dimension of 1.53–1.19 nm) may act as visitor molecules unfold into the hole inside in doses ample by the mesoporous shells. Moreover, positively charged DOX may bind the negatively charged Fe-PHCN to manufacture the Fe-PHCN@DOX by way of electrostatic interactions. As proven in Fig. 2H, the dimensions of the Fe-PHCN@DOX was roughly 204 nm with out an apparent enhance in dimension relative to the unique Fe-PHCN. TEM pictures confirmed that some morphologically inhomogeneous agglomerates had been noticed within the hole inside of Fe-PHCN@DOX in comparison with that noticed for the Fe-PHCN (Fig. 2I), which is likely to be attributed to the DOX loaded into the hole core. From UV–vis–NIR absorption spectra (Fig. 2J), Fe-PHCN@DOX exhibited new absorption peak at close to 480 nm after the combination of the DOX. The outcomes had been additionally validated from an evident change of resolution shade earlier than and after DOX loading (Extra file 1: Fig. S8). Due to this fact, these outcomes demonstrated DOX profitable loading into the nano-nuclear-reactors. The loading effectivity of the DOX within the nano-nuclear-reactors was roughly 36.67 wt%, as decided by the drug loading experiment. Moreover, the steadiness of the Fe-PHCN@DOX nano-nuclear-reactors in numerous options, together with water, PBS, and DMEM medium containing 10% FBS, was additional evaluated for six days by DLS measurement. It was noticed that no apparent dimension modifications of the Fe-PHCN@DOX within the numerous options (Extra file 1: Fig. S9), demonstrating the wonderful stability of the nano-nuclear-reactors in water and physiological resolution.
Photothermal impact and pH/NIR accountable drug launch
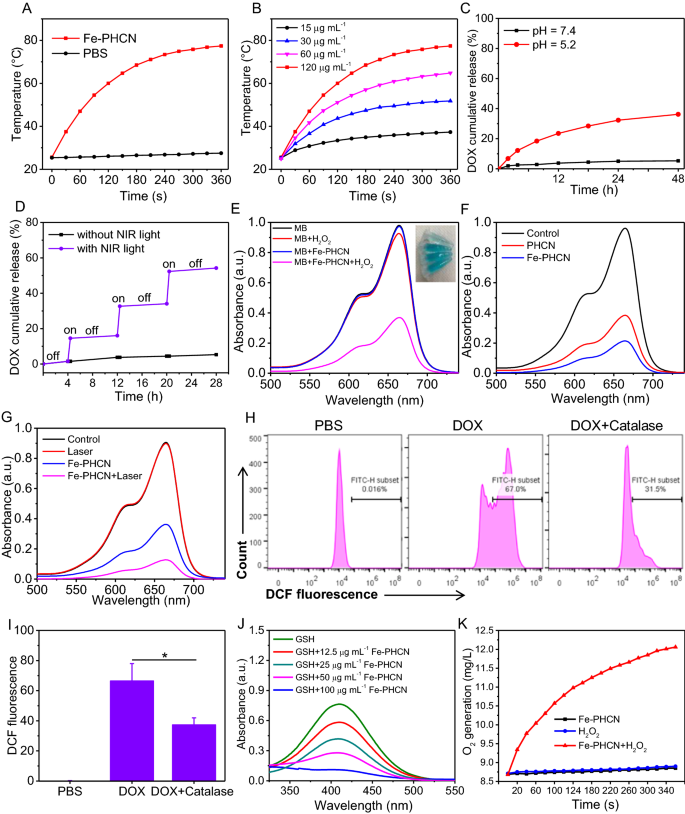
Primarily based on the sturdy NIR absorption, the photothermal properties of the nano-nuclear-reactors had been investigated by monitoring the temperature modifications underneath NIR laser irradiation utilizing an infrared thermal imaging digital camera. As proven in Fig. 3A, temperature of the Fe-PHCN options markedly elevated underneath 1064 nm laser irradiation at 1 W cm−2 for six min, whereas no apparent temperature modifications of PBS had been noticed. Furthermore, the Fe-PHCN confirmed wonderful concentration-dependent photothermal properties underneath laser irradiation (Fig. 3B). These outcomes confirmed that the wonderful photothermal effectivity of the Fe-PHCN may enable it to be an environment friendly photothermal agent. DOX launch behaviors from Fe-PHCN@DOX at completely different pH values (7.4 and 5.2) had been investigated. Fe-PHCN@DOX displayed sustained drug launch habits (Fig. 3C), and the quantity of DOX launched elevated with reducing pH on account of lowered electrostatic interactions between the DOX and the Fe-PHCN in acidic pH circumstances. As proven in Fig. 3D, solely a small quantity of DOX was noticed with out NIR gentle, whereas a burst launch of DOX occurred underneath NIR gentle irradiation. The additional elevated DOX launch underneath repeated laser irradiation was attributed to the photothermal impact of the Fe-PHCN, thus producing warmth to advertise drug diffusion.
A number of properties of Fe-PHCN@DOX nano-nuclear-reactors. A Temperature elevation curves of PBS and Fe-PHCN uncovered to a 1064 nm NIR laser for six min. B Temperature elevation curves of Fe-PHCN with completely different concentrations underneath laser irradiation. Launch curves of DOX from Fe-PHCN@DOX at completely different pH (C) and with or with out NIR irradiation (D). E UV − vis spectra of MB aqueous resolution from completely different teams. F UV − vis spectra of MB aqueous resolution with Fe-PHCN and PHCN remedy. G UV − vis spectra of MB in Fe-PHCN resolution handled with/with out laser irradiation. H, I Intracellular ROS content material was analyzed underneath DOX and DOX plus Catalase remedies utilizing DCF as a ROS sensor. J GSH depletion underneath completely different concentrations of Fe-PHCN. (Ok) O2 technology of Fe-PHCN underneath with/with out H2O2 remedy. The p values had been analyzed utilizing the Log-rank (Mantel-Cox) take a look at. Information are introduced because the imply ± normal error of the imply. *p < 0.05, **p < 0.01, ***p < 0.001
Nanocatalytic exercise, GSH depletion, and O2 technology of Fe-PHCN nano-nuclear-reactors
To additional examine nanocatalytic properties, the •OH technology of Fe-PHCN was decided utilizing the methylene blue (MB) degradation methodology [47]. No important modifications of MB absorption had been noticed in H2O2 or Fe-PHCN in comparison with Fe-PHCN plus H2O2 (Fig. 3E), indicating that the Fe-PHCN may catalyze H2O2 to generate •OH. The nanocatalytic properties of Fe-PHCN in a concentration-dependent method had been confirmed (Extra file 1: Fig. S10). As proven in Fig. 3F, greater •OH technology by Fe-PHCN was discovered in comparison with PHCN, suggesting that the introduction of Fe3+ enhancing nanocatalytic effectivity [39]. Fe-PHCN underneath the 1064 nm laser irradiation may enhance •OH technology (Fig. 3G), which revealed that photothermal impact mediated excessive temperature may enhance the nanocatalytic impact [48, 49]. Moreover, DOX contributing to producing H2O2 in most cancers cells was evaluated utilizing the sensor 2ʹ,7ʹ-dichlorofluorescein diacetate (DCF) [50]. As proven in Fig. 3H, I, excessive inexperienced fluorescence was noticed in cells after DOX remedy, suggesting excessive H2O2 stage. In distinction, the cells handled with Catalase, a H2O2 scavenger, confirmed comparatively low ROS stage. These outcomes demonstrated that DOX can generate H2O2 in most cancers cells, doubtlessly enhancing nanocatalytic effectivity. Moreover, the GSH depletion properties of Fe-PHCN had been investigated utilizing the GSH equipment. As proven in Fig. 3J, the content material of GSH was step by step decreased with the growing concentrations of Fe-PHCN. The outcomes revealed that Fe-PHCN exhibited wonderful GSH depletion potential. The catalase-like exercise of Fe-PHCN was investigated by monitoring O2 manufacturing utilizing a dissolved oxygen meter. Larger O2 technology was noticed handled with Fe-PHCN plus H2O2, whereas no apparent O2 manufacturing handled with Fe-PHCN had been noticed (Fig. 3Ok), which demonstrated that the Fe-PHCN may successfully generate O2 to alleviate tumor hypoxia.
Mobile uptake and therapeutic efficacy
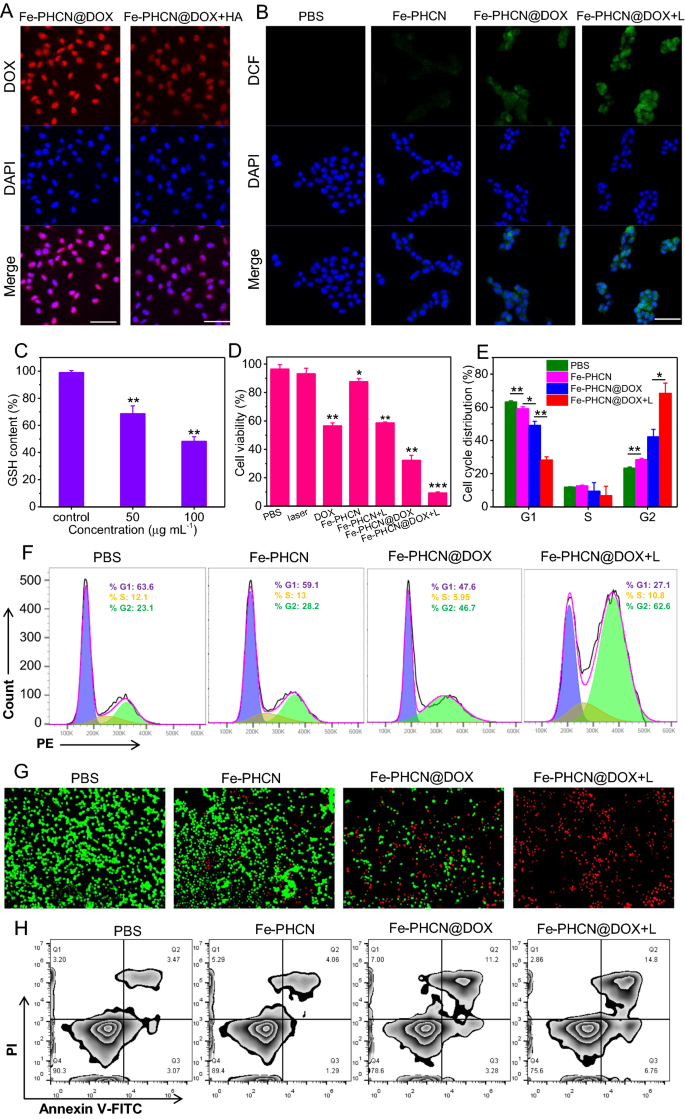
The intracellular distribution and uptake of the Fe-PHCN@DOX nano-nuclear-reactors had been evaluated by confocal microscopy and circulate cytometry, respectively. As proven in Fig. 4A, the presence of free HA may considerably inhibit the intracellular distribution of the Fe-PHCN@DOX due to the aggressive interplay between Fe-PHCN@DOX and HA on CD44 receptors on CT26 cells [51]. The circulate cytometric outcomes had been in keeping with these from confocal microscopy (Extra file 1: Fig. S11), suggesting the HA-mediated focusing on property of the Fe-PHCN@DOX. To review mobile ROS manufacturing of the Fe-PHCN@DOX nano-nuclear-reactors, a fluorescent probe DCF was used. Highest inexperienced fluorescent depth was noticed in cells handled with Fe-PHCN@DOX plus laser irradiation in comparison with different teams (Fig. 4B; Extra file 1: Fig. S12), suggesting that Fe-PHCN@DOX may generate extra •OH in cells by introducing Fe3+/DOX and utilizing laser irradiation. These outcomes confirmed that the Fe-PHCN@DOX nano-nuclear-reactors displayed superior nanocatalytic efficacy in most cancers cells. In the meantime, the dramatic depletion of intracellular GSH was detected in most cancers cells handled with the Fe-PHCN@DOX nano-nuclear-reactors (Fig. 4C). This discovering revealed that Fe-PHCN@DOX exhibited wonderful intracellular GSH depletion potential, doubtlessly enhancing the nanocatalytic impact by destroying mobile antioxidant protection system. The therapeutic efficacy of the Fe-PHCN@DOX nano-nuclear-reactors was evaluated by a CCK-8 assay. As proven in Fig. 4D, the Fe-PHCN@DOX underneath laser irradiation induced a remarkably greater cell toxicity of 90.54%, far greater than that of different teams. This consequence revealed that Fe-PHCN@DOX may show optimum anticancer impact by photothermal impact, •OH technology, GSH depletion, and DOX-induced cytotoxicity. Moreover, cell cycle development was analyzed utilizing PI staining. The upper proportion of cells handled with Fe-PHCN@DOX plus laser (62.6%) within the G2 section was noticed than that of PBS (23.1%), Fe-PHCN (28.2%), and Fe-PHCN@DOX (46.7%) (Fig. 4E, F), which advised that the Fe-PHCN@DOX nano-nuclear-reactors may induce cell cycle arrest within the G2 section. Furthermore, fluorescence staining of the dwelling/lifeless cells additionally confirmed that the Fe-PHCN@DOX nano-nuclear-reactors underneath laser irradiation precipitated extra cell demise as in contrast with that in different teams (Fig. 4G). Annexin V-FITC/PI cell co-staining (Fig. 4H; Extra file 1: Fig. S13) additional confirmed that the Fe-PHCN@DOX nano-nuclear-reactors displayed the efficient anticancer efficacy underneath laser irradiation.
Mobile uptake and therapeutic efficacy of Fe-PHCN@DOX nano-nuclear-reactors. A Confocal pictures of the CT26 subcellular localization of the Fe-PHCN@DOX with/with out HA remedy. The dimensions bar represents 50 μm. B Fluorescence pictures of DCF-stained CT26 cells underneath completely different group remedy. The dimensions bar represents 50 μm. C Intracellular GSH depletion underneath completely different concentrations of Fe-PHCN@DOX. D Cell survival price after completely different group remedy by the CCK-8 assay. E, F Cell cycle arrest in G2 section was measured underneath completely different group remedy. G Fluorescence pictures of calcein AM (inexperienced, dwell cells) and PI (crimson, lifeless cells) costained CT26 cells after completely different group remedy. The dimensions bar represents 100 μm. H Circulate cytometry evaluation of apoptosis in CT26 cells handled with completely different teams. The p values had been analyzed utilizing the Log-rank (Mantel-Cox) take a look at. Information are introduced because the imply ± normal error of the imply. *p < 0.05, **p < 0.01, ***p < 0.001
Nanobody binding exercise, mobile O2 manufacturing, and ICD induction
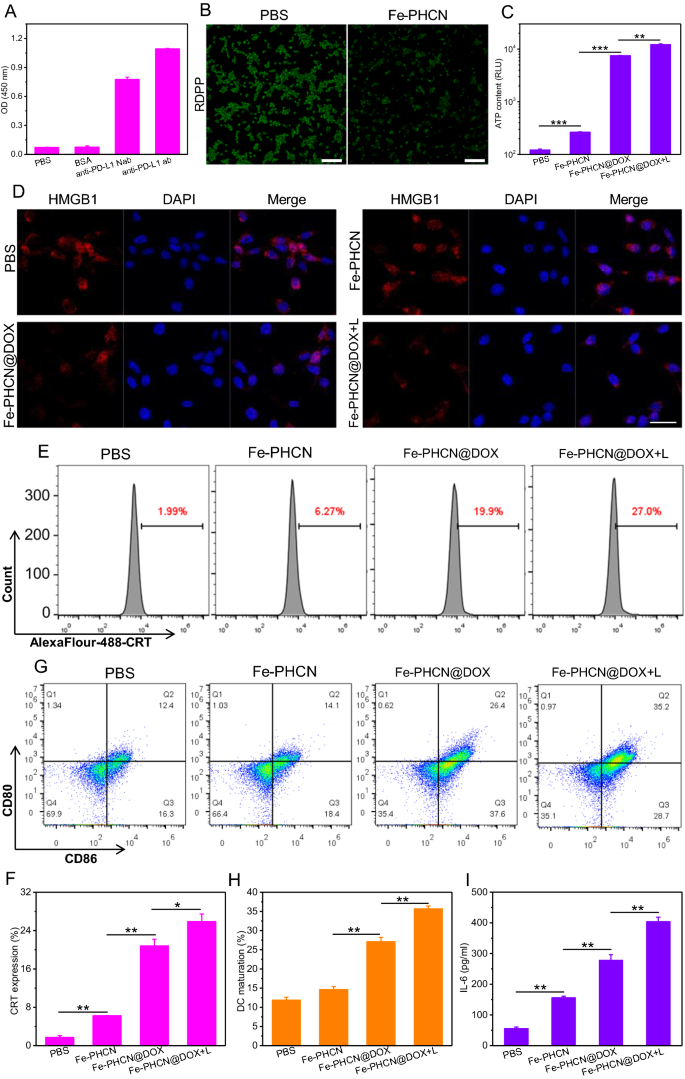
To confirm our speculation that the nano-nuclear-reactors induced efficient immune activation of T cells by reversing immunosuppression in low immunogenicity and hypoxia tumor, we detect the PD-L1/anti-PD-L1 nanobody binding exercise, the O2 manufacturing, and ICD impact in vitro. The molecular weight of anti-PD-L1 nanobody was considerably decrease than that of the anti-PD-L1 antibody in protein gel electrophoresis (Extra file 1: Fig. S14), confirming the profitable preparation of the nanobody. Anti-PD-L1 nanobody exhibited stronger tissue permeability and distribution in tumor tissue than that of economic anti-PD-L1 monoclonal antibody (Extra file 1: Fig. S15). Determine 5A confirmed that the anti-PD-L1 nanobody/PD-L1 antigen binding exercise was just like the business antibody. Furthermore, the anti-PD-L1 nanobody may successfully bind the PD-L1 receptor on CT26 cells (Extra file 1: Fig. S16). These outcomes indicated that the anti-PD-L1 nanobody can blockade the PD-1/PD-L1 immunosuppressive pathway. The degrees of TNF-α and IFN-γ launched from T cells had been clearly elevated underneath anti-PD-L1 nanobody remedy (Extra file 1: Fig. S17), suggesting nanobody-mediated PD-L1 blockade selling T cell activation. We additional measured PD-L1 blockade-mediated T cell activation by staining the activation maker CD69. The share of CD69+CD8+ T cells was considerably elevated underneath anti-PD-L1 nanobody remedy (Extra file 1: Fig. S18), suggesting nanobody-mediated PD-L1 blockade selling T cell activation. The mobile O2-generating actions of Fe-PHCN utilizing the O2 probe [Ru(dpp)3]Cl2 (RDPP). As proven in Fig. 5B, in comparison with the PBS group, decrease inexperienced fluorescence of RDPP was noticed in cells handled with Fe-PHCN. Moreover, the elevated M1-type macrophages (CD11b+F4/80+CD86+CD206−) and decreased M2-type macrophages (CD11b+F4/80+CD86−CD206+) had been noticed underneath Fe-PHCN remedy (Extra file 1: Fig. S19). The outcomes demonstrated that the Fe-PHCN may successfully produce O2, thus doubtlessly polarizing M2-type macrophages into M1-type macrophages. To judge the impact of the Fe-PHCN@DOX nano-nuclear-reactors-induced ICD, we chosen calcium netting protein (CRT), adenosine triphosphate (ATP), and excessive mobility group field 1 (HMGB1) as indicators [52, 53]. In contrast with PBS group, the elevated ATP and CRT stage, and decreased HMGB1 stage in nuclei with Fe-PHCN and Fe-PHCN@DOX remedies was noticed (Fig. 5C–F). In distinction, the group handled with Fe-PHCN@DOX plus laser irradiation induced considerably elevated ATP secretion, CRT expression, and HMGB1 launch from nuclei, suggesting that nano-nuclear-reactors may function a wonderful ICD amplifier. The DAMPs launched from CT26 tumor cells present process ICD had been introduced to immatured DCs for selling DC maturation (CD11c+CD86+CD80+), which is accountable for activating T cell immune responses. DC2.4 cells had been chosen for testing in vitro DCs maturation. The CT26 cells had been pre-treated with nano-nuclear-reactors after which co-cultured immature DC2.4 cells. The frequency of matured DC2.4 cells was then analyzed by circulate cytometry. In contrast with PBS group (12.4%), the reasonably elevated DCs maturation (CD11c+CD86+CD80+) with Fe-PHCN (14.1%) and Fe-PHCN@DOX (26.4%) remedy was noticed (Fig. 5G, H; Extra file 1: Fig. S20). Remarkably, the Fe-PHCN@DOX plus laser irradiation considerably promoted DCs maturation (35.2%). These outcomes demonstrated that the Fe-PHCN@DOX nano-nuclear-reactors based mostly ICD impact effectively promoted DCs maturation for evoking T cell immune activation. Furthermore, the secretion ranges of pro-inflammatory cytokines IL-6 and TNF-α had been investigated utilizing enzyme-linked immunosorbent assay (ELISA). In contrast with different teams, the secretion of IL-6 and TNF-α noticed elevated underneath Fe-PHCN@DOX remedy (Fig. 5I; Extra file 1: Fig. S21), which additional promoted T cell activation.
Nanobody binding exercise, mobile O2 manufacturing, and ICD induction of Fe-PHCN@DOX nano-nuclear-reactors. A The binding exercise of PD-L1 antigen with BSA, anti-PD-L1 nanobody (Nab), and anti-PD-L1 antibody (ab) was measured by ELISA. B O2 technology in CT26 cells handled with completely different teams. The dimensions bar is 100 μm. The ATP content material (C), the HMGB1 stage in nuclei (D), and the CRT expression on the floor of CT26 cells (E, F) after completely different group remedy. G, H Quantification of CD80 and CD86 expression on the floor of DC2.4 cells after completely different remedy by circulate cytometry. I ELISA evaluation of the degrees of cytokines IL-6 secreted by DC2.4 cells within the medium. The dimensions bar is 25 μm. The p values had been analyzed utilizing the Log-rank (Mantel-Cox) take a look at. Information are introduced because the imply ± normal error of the imply. *p < 0.05, **p < 0.01, ***p < 0.001
In vivo biodistribution and tumor microenvironment alterations by nano-nuclear-reactors
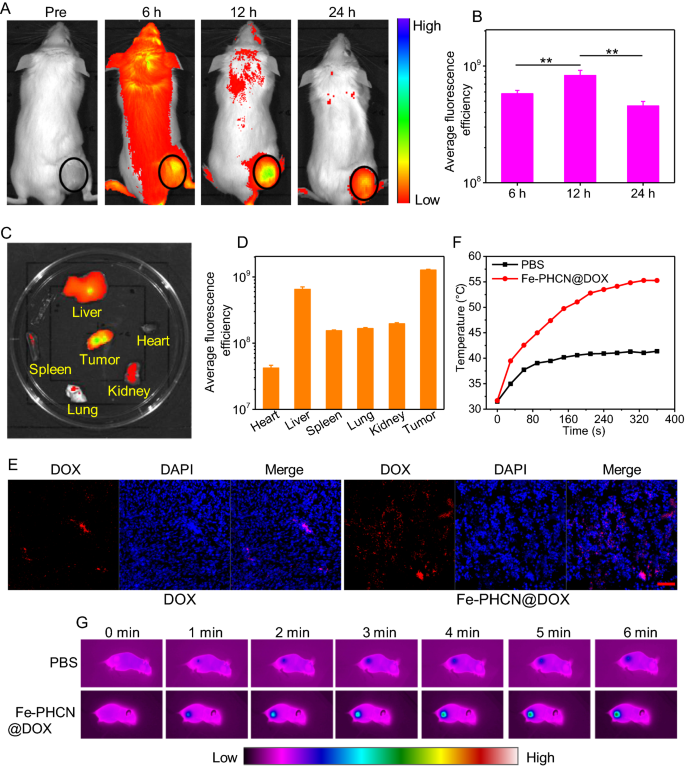
Excessive accumulation of nano-nuclear-reactors in tumors is a prerequisite for attaining enhanced antitumor therapeutic outcomes and decrease facet impact to tissues. To visualise the dynamic distribution of nano-nuclear-reactors in vivo, a NIR dye Chlorin e6 (Ce6) was included into Fe-PHCN@DOX to generate Ce6-loaded Fe-PHCN@DOX (Fe-PHCN@DOX/Ce6) [50]. Tumor-bearing mice had been intravenously injected with Fe-PHCN@DOX/Ce6, and the biodistribution was monitored at completely different time intervals utilizing an in vivo imaging system. The fluorescence indicators of Fe-PHCN@DOX/Ce6 within the tumor area elevated with growing time and reached a peak at 12 h after injection, and step by step decreased at 24 h (Fig. 6A, B). This discovering revealed that the Fe-PHCN@DOX/Ce6 may effectively enrich within the tumor area for a chronic time. Furthermore, direct ex vivo fluorescence imaging of excised tumors obtained at 24 h (Fig. 6C, D), confirmed the efficient distribution of Fe-PHCN@DOX/Ce6 in tumor tissue, suggesting that HA focusing on property considerably enhanced the tumor accumulation of the nano-nuclear-reactors. In the meantime, the buildup of DOX in tumors was additionally investigated after Fe-PHCN@DOX injection. The confocal pictures confirmed that Fe-PHCN@DOX considerably enhanced the fluorescent indicators of DOX in tumors as in contrast with free DOX, which additional confirmed successfully enhanced DOX accumulation in tumor tissues by Fe-PHCN@DOX nano-nuclear-reactors (Fig. 6E). Furthermore, the dramatic lower of GSH in tumor was noticed with the growing focus of Fe-PHCN@DOX (Extra file 1: Fig. S22), which indicated that the nano-nuclear-reactors may regulate the TME. Moreover, to watch thermal dynamics in tumor handled with the Fe-PHCN@DOX nano-nuclear-reactors after i.v. injection, photothermal imaging could possibly be used to check the temperature variation of tumor by an infrared thermal digital camera. As proven in Fig. 6F, G, the temperature of the Fe-PHCN@DOX within the tumor area step by step elevated with growing laser irradiation time, and the temperature considerably elevated to 55.27 °C inside 6 min. In distinction, the temperature of the tumor handled with PBS was not remarkably elevated. Due to this fact, this consequence confirmed that the Fe-PHCN@DOX nano-nuclear-reactors may act as a wonderful photothermal agent in vivo.
In vivo biodistribution and tumor microenvironment alterations by nano-nuclear-reactors. A In vivo fluorescence imaging of CT26 tumor-bearing mice intravenously injected with Fe-PHCN@DOX/Ce6 recorded underneath completely different time factors. B The imply fluorescence depth on the tumor websites was quantified utilizing CRi maestro in vivo imaging system. Ex vivo fluorescence pictures (C) and the imply fluorescence depth (D) of main organs and tumor dissected from mice injected with Fe-PHCN@DOX/Ce6. E The fluorescent sign of free DOX and Fe-PHCN@DOX in tumor tissues was recorded utilizing confocal microscopy. The dimensions bar represents 50 μm. Temperature elevation curves (F) and infrared photothermal pictures (G) of tumor-bearing mice measured after intravenous injection of PBS and Fe-PHCN@DOX plus laser irradiation for six min. The p values had been analyzed utilizing the Log-rank (Mantel-Cox) take a look at. Information are introduced because the imply ± normal error of the imply. *p < 0.05, **p < 0.01, ***p < 0.001
Enhanced therapeutic efficacy on CT26 tumor in vivo
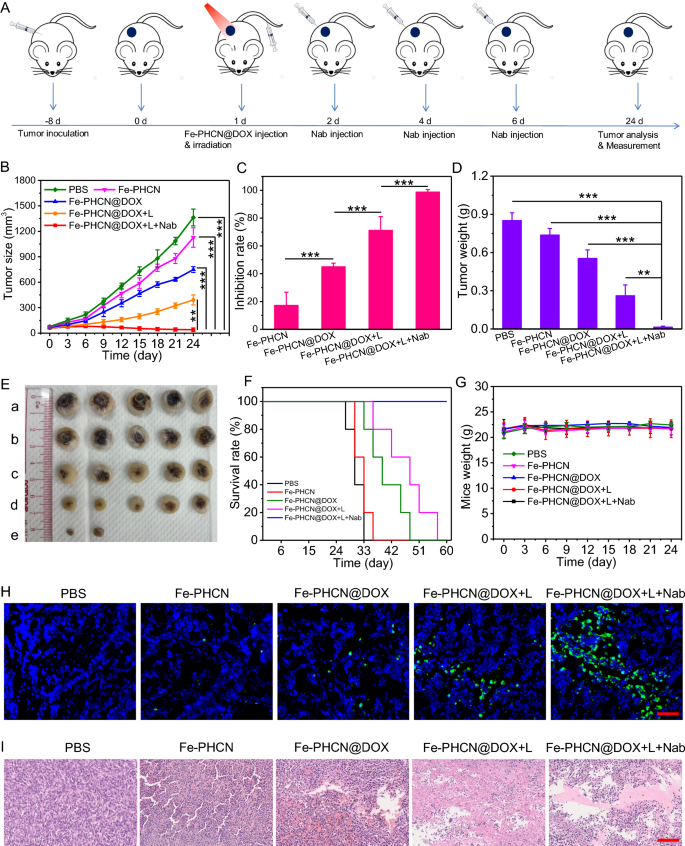
Motivated by the efficient tumor accumulation and wonderful photothermal efficiency in tumor of the Fe-PHCN@DOX nano-nuclear-reactors, we subsequent evaluated the antitumor therapeutic potential. The xenograft CT26 tumor fashions had been established, and tumor-bearing mice had been randomly divided into 5 teams: (a) PBS; (b) Fe-PHCN; (c) Fe-PHCN@DOX; (d) Fe-PHCN@DOX plus laser irradiation; and (e) Fe-PHCN@DOX plus laser irradiation plus anti-PD-L1 nanobody. As proven in Fig. 7A, after 8 days’ tumor inoculation, the tumor quantity attain about 60 mm3, after which tumor-bearing mice had been intravenously injected with nano-nuclear-reactors on day 1. Then, tumors had been carried out underneath 1064 nm laser irradiation 12 h post-injection, and anti-PD-L1 nanobody was intra-tumorally injected on day 2, 4, and 6. Though systemic remedy administration of immune checkpoint inhibitors is typical approaches, their serum pharmacokinetics is unpredictable. Notably, antibody penetration from the circulation into stable tumors is restricted. In the meantime, systemic distribution might increase questions of safety. Systemic irritation usually prevents the usage of efficacious doses of immune checkpoint inhibitors [54]. Intratumoral supply is a lovely choice to extend the bioavailability of nanobody in TME [55, 56]. Within the preliminary in vivo experiment, the Fe-PHCN@DOX displayed greater inhibition impact of tumor development than that of PBS and free DOX (Extra file 1: Fig. S23). Tumor volumes had been monitored each 3 days. In contrast with the PBS group, the Fe-PHCN (17.3%) and Fe-PHCN@DOX (45.05%) teams exhibited the partly inhibition impact of tumor development (Fig. 7B). In distinction, the Fe-PHCN@DOX group may induce the upper tumor suppression impact (71.29%) plus laser irradiation remedy. Furthermore, the group of Fe-PHCN@DOX plus laser plus anti-PD-L1 nanobody resulted in a strongest tumor suppressing impact (98.82%), which attributable to the improved antitumor therapeutic impact (Fig. 7C). In the meantime, the group of Fe-PHCN@DOX plus laser plus anti-PD-L1 nanobody may successfully cut back tumor weight (Fig. 7D), virtually ablate the tumor dimension (Fig. 7E), and considerably extended the survival charges of the mice (Fig. 7F) as in contrast with different teams. No important variations in mice weight had been noticed in any of the teams in the course of the remedy course of, indicating the wonderful biosafety of the Fe-PHCN@DOX nano-nuclear-reactors (Fig. 7G). Moreover, to judge the antitumor therapeutic consequence, hematoxylin and eosin (H&E) and terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) staining was used to research the tumors. In contrast with different teams, Fe-PHCN@DOX underneath laser irradiation along with anti-PD-L1 nanobody may induce outstanding tumor tissue harm (Fig. 7H) and extra apoptosis/necrosis of tumors (Fig. 7I). Taken collectively, these outcomes demonstrated that the Fe-PHCN@DOX nano-nuclear-reactors plus PD-L1 immune checkpoint blockade may successfully enhance the antitumor therapeutic effectiveness.
Antitumor therapeutic efficacy of on CT26 tumor in vivo. A Remedy schedule for Fe-PHCN@DOX and anti-PD-L1 nanobody (Nab) mixture remedy. Tumor development curves (B) and inhibition charges (C) from numerous teams, together with a) PBS; b) Fe-PHCN; c) Fe-PHCN@DOX; d) Fe-PHCN@DOX plus laser; and e) Fe-PHCN@DOX plus laser plus anti-PD-L1 nanobody. Tumor weights (D) and consultant pictures of tumor dimension (E) of mice in every group. Survival charges (F) and physique weights (G) of mice after completely different remedies. H Fluorescence pictures of TUNEL-stained tumor slices after numerous remedies. Cell nuclei had been stained with DAPI (blue fluorescence). Inexperienced fluorescence signifies TUNEL-positive cells. I H&E-stained pictures of tumor slices obtained from completely different teams of mice. The dimensions bar represents 100 μm. The p values had been analyzed utilizing the Log-rank (Mantel-Cox) take a look at. Information are introduced because the imply ± normal error of the imply. *p < 0.05, **p < 0.01, ***p < 0.001
In vivo ICD induction and immunosuppressive TME reprogramming
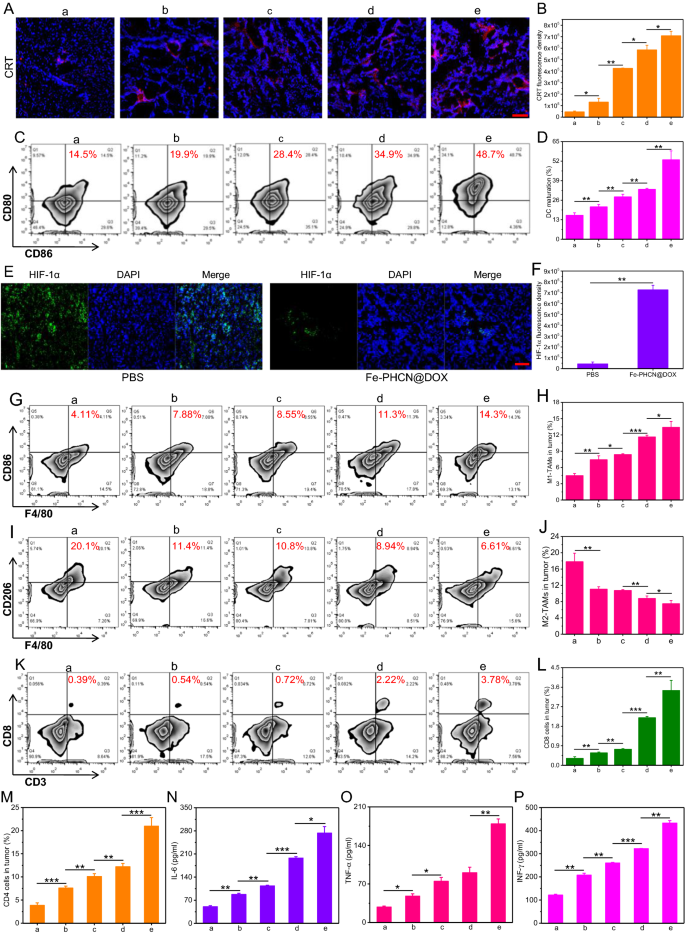
On the premise of the wonderful antitumor efficacy, the potential mechanism that nano-nuclear-reactors and nanobody mediated persistently amplifying T cell immune response was explored in vivo. Firstly, the ICD impact in tumors and DCs maturation in lymph nodes had been investigated. As proven in Fig. 8A, B, remarkably greater CRT expression was noticed in tumor tissue handled with Fe-PHCN@DOX plus laser plus nanobody group as in contrast with different teams, suggesting their superior capability in inducing potent ICD impact for the in vivo DCs maturation. As anticipated, the Fe-PHCN@DOX plus laser plus nanobody group may successfully promote DCs maturation (CD11c+CD86+CD80+) with greater share of matured DCs (48.7%) than that of PBS (14.5%), Fe-PHCN (19.9%), Fe-PHCN@DOX (28.4%), and Fe-PHCN@DOX plus laser (34.9%) teams (Fig. 8C, D; Extra file 1: Fig. S24). Due to this fact, after tumor apoptosis induced by the ICD impact, the launched DAMPs could possibly be introduced to immatured DCs to successfully simulate DCs maturation, thereby doubtlessly stimulating T cell activation. Secondly, the hypoxic situation and macrophages polarization in TME had been analyzed. From immunofluorescent staining pictures, the excessive inexperienced fluorescence indicators of hypoxia inducible factors-1 alpha (HIF-1α) in tumors handled with PBS was noticed, whereas the decrease inexperienced fluorescence indicators of HIF-1α in tumors handled with the Fe-PHCN@DOX (Fig. 8E, F). This discovering revealed that Fe-PHCN@DOX nano-nuclear-reactors can successfully relieve hypoxia in tumors, doubtlessly polarizing towards M1-type macrophage. Extra M1-type macrophage (CD11b+F4/80+CD86+) share (14.3%) in tumors of mice handled with the Fe-PHCN@DOX plus laser plus nanobody group was noticed as in contrast with PBS (4.11%), Fe-PHCN (7.88%), Fe-PHCN@DOX (8.55%), and Fe-PHCN@DOX plus laser (11.3%) teams (Fig. 8G, H; Extra file 1: Fig. S25A). The Fe-PHCN@DOX plus laser plus nanobody group additionally successfully decreased the proportion of M2-type macrophages (CD11b+F4/80+CD206+) in tumors (Fig. 8I, J; Extra file 1: Fig. S25B). These outcomes confirmed sturdy proof that the polarization of M2-type macrophages to M1-type macrophages by relieving tumor hypoxia, thus doubtlessly enhancing T cell immune operate. Lastly, T cell-mediated mobile immunity together with the activation and infiltration of CD8+ cytotoxic T (CD3+CD8+) cells and CD4+ helper T (CD3+CD4+) cells in TME was investigated. The best share of CD8+ cytotoxic T cells (3.78%) in tumors in Fe-PHCN@DOX plus laser plus nanobody group was discovered when put next with PBS (0.39%), Fe-PHCN (0.54%), Fe-PHCN@DOX (0.72%), and Fe-PHCN@DOX plus laser (2.22%) teams (Fig. 8Ok, L; Extra file 1: Fig. S26A). In line with CD8+ cytotoxic T cells, the Fe-PHCN@DOX plus laser plus nanobody group additionally remarkably elevated the very best share of CD4+ helper T cells (Fig. 8M; Extra file 1: Fig. S26B, S27). These outcomes demonstrated the triggering efficient T cell activation and infiltration in TME by ICD impact, macrophages polarization, and nanobody blockade. To additional research the anti-tumor mechanism of CD8+ and CD4+ T cells, we decided the proportion of IFN-γ+, Granzyme B+ and Perforin+ in CD8+ T cells in addition to IFN-γ+ and TNF-α+ in CD4+ cells remoted from tumor tissues. The group of Fe-PHCN@DOX plus laser plus anti-PD-L1 nanobody may set off greater share of IFN-γ+, Granzyme B+, Perforin+ CD8+ T cells (Extra file 1: Fig. S28), and IFN-γ+, TNF-α+ CD4+ cells (Extra file 1: Fig. S29) in tumor tissue than that of different group. In comparison with different remedy, the decrease share of Treg cells (CD3+CD4+Foxp3+) in tumor tissue was noticed after the Fe-PHCN@DOX plus laser plus nanobody remedy (Extra file 1: Fig. S30). Importantly, Fe-PHCN@DOX plus laser plus nanobody confirmed the next variety of infiltrating T cells in tumors than that of different group (Extra file 1: Fig. S31). The above outcomes indicated that nano-nuclear-reactors mixed with nanobody may successfully activate and strengthen CD8+ cytotoxic T cells and CD4+ helper T cells, thus considerably enhancing the anti-tumor efficacy. Moreover, the degrees of cytokines together with IL-6, TNF-α, and interferon-γ (IFN-γ) within the blood had been evaluated. The upper ranges of those cytokines had been discovered after the Fe-PHCN@DOX plus laser plus nanobody remedy than these of different teams (Fig. 8N–P). Collectively, the above outcomes indicated that nano-nuclear-reactors along with nanobody promoted the ICD-mediated DCs maturation, macrophages polarization, and anti-PD-L1 blockade for efficient antitumor T cell immune response by reversing the low immunogenicity, hypoxia, and immunosuppressive TME.
In vivo immunogenic cell demise induction and immunosuppressive TME reprogramming. Confocal microscopy pictures (A) and fluorescence depth (B) of CRT protein publicity in tumor tissues handled with a) PBS; b) Fe-PHCN; c) Fe-PHCN@DOX; d) Fe-PHCN@DOX plus laser; and e) Fe-PHCN@DOX plus laser plus anti-PD-L1 nanobody. The quantification (C) and share (D) of matured DCs cells (CD11c+CD86+CD80+) by circulate cytometric analyses after numerous remedies. Consultant immunofluorescence pictures (E) and relative quantitative evaluation (F) of hypoxia areas in tumors handled with completely different teams after staining with DAPI (blue) and anti-HIF-1α antibody (inexperienced). The quantification (G) and share (H) of M1-type macrophages (CD11b+F4/80+CD86+) by circulate cytometric analyses after numerous remedies. The quantification (I) and share (J) of M2-type macrophages (CD11b+F4/80+CD206+) by circulate cytometric analyses after numerous remedies. The quantification (Ok) and share (L) of CD8+ cytotoxic T (CD3+CD8+) cells by circulate cytometric analyses after numerous remedies. M The share of CD4+ helper T (CD3+CD4+) cells by circulate cytometric analyses after numerous remedies. N–P ELISA evaluation of the degrees of cytokines IL-6, TNF-⍺, and IFN-γ in serum of mice after numerous remedies. The dimensions bar represents 50 μm. The p values had been analyzed utilizing the Log-rank (Mantel-Cox) take a look at. Information are introduced because the imply ± normal error of the imply. *p < 0.05, **p < 0.01, ***p < 0.001
Abscopal impact and metastasis prevention
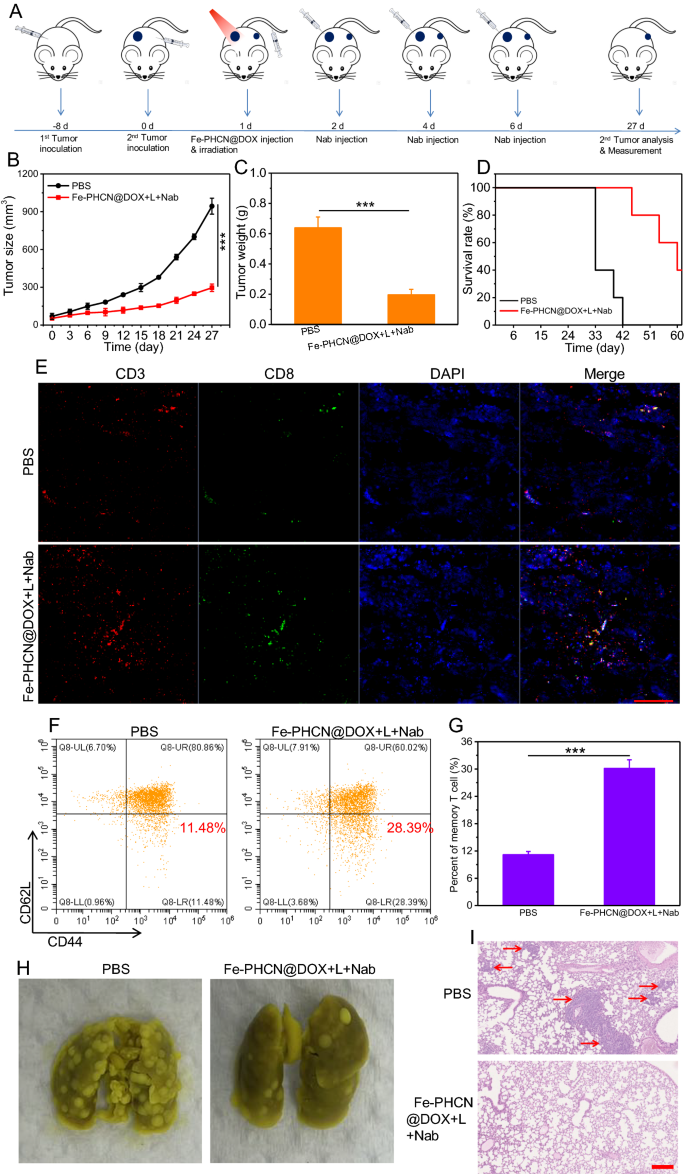
An essential characteristic of immune techniques is the flexibility of immune reminiscence that protects organisms from tumor cells re-attacking and tumor metastasis. To additional consider the immune reminiscence results of nano-nuclear-reactors along with nanobody, CT26 cells had been seeded on the left/proper facet of the identical mice on day − 8 and 0 (Fig. 9A). After the tumor quantity attain about 60 mm3, and tumor-bearing mice had been randomly divided into two teams: a) PBS; b) Fe-PHCN@DOX plus laser plus anti-PD-L1 nanobody. Then, tumor-bearing mice had been intravenously injected with nano-nuclear-reactors on day 1. Then, the first tumors had been carried out underneath 1064 nm laser irradiation 12 h post-injection, and anti-PD-L1 nanobody was intratumorally injected on day 2, 4, and 6. For major tumors, virtually full elimination of the tumors with Fe-PHCN@DOX plus laser plus nanobody remedy was discovered, in keeping with the above outcomes. For distant tumors, the tumors quantity was monitored by caliper measurement. As confirmed in Fig. 9B, the expansion charges of distant tumors handled with PBS group had been quickly, whereas important development inhibition charges of distant tumors handled with Fe-PHCN@DOX plus laser plus nanobody group had been noticed. This discovering indicated that nano-nuclear-reactors along with nanobody mediated nano-immunotherapy may set off sturdy immune responses. Furthermore, the Fe-PHCN@DOX plus laser plus nanobody group may successfully cut back the tumor weight and lengthen the mice survival charges as in contrast with the PBS group (Fig. 9C, D). From immunofluorescent staining pictures, a big enhance of infiltrating CD8+ T cell in distant tumors in Fe-PHCN@DOX plus laser plus nanobody group was additionally noticed in comparison with that of the PBS group (Fig. 9E), suggesting strong triggering systemic T cell immune responses. To additional make clear the underlying mechanisms of the persistently amplifying immune responses, we evaluated the effector reminiscence T cells (CD3+CD8+CD44+CD62L−) within the spleen. The share of the effector reminiscence T cells within the Fe-PHCN@DOX plus laser plus nanobody group was a lot greater than that of the PBS group (Fig. 9F, G; Extra file 1: Fig. S32). In line with the outcomes of effector reminiscence T cells, the degrees of cytokines IFN-γ noticed elevated after Fe-PHCN@DOX plus laser plus nanobody remedy (Extra file 1: Fig. S33). Our information strongly evidenced that nano-nuclear-reactors along with nanobody may induce a long-term immune reminiscence impact. Inspired by the sturdy immune reminiscence in opposition to the distant tumors, we subsequently investigated the anti-metastasis results in a CT26 lung metastasis mannequin. Extra lung metastasis nodules had been noticed mice in lung handled with the PBS group, whereas mice handled with nano-nuclear-reactors along with nanobody group confirmed much less lung metastasis (Fig. 9H). This consequence additionally confirmed by H&E staining lung slices (Fig. 9I). Taken collectively, our outcomes demonstrated that Fe-PHCN@DOX nano-nuclear-reactors along with nanobody may successfully promote persistently systemic antitumor T cell immune responses and inhibit distant and metastatic tumors.
Abscopal impact and lung metastasis prevention in vivo. A Remedy schedule for Fe-PHCN@DOX and anti-PD-L1 nanobody mixture remedy. Tumor development curves (B) and tumor weights (C) from completely different teams. D Survival charges of mice after completely different remedies. E CLSM pictures of CD3+CD8+ T cells after staining with DAPI (blue), anti-CD3 antibody (crimson) and anti-CD8 antibody (inexperienced), respectively. The dimensions bar is 100 µm. The quantification (F) and share (G) of the effector reminiscence T cells (CD3+CD8+CD44+CD62L−) by circulate cytometric analyses after completely different remedies. H Consultant pictures of the lung metastatic nodules. I H&E staining of lungs after completely different remedies. The dimensions bar is 200 μm. The p values had been analyzed utilizing the Log-rank (Mantel-Cox) take a look at. Information are introduced because the imply ± normal error of the imply. *p < 0.05, **p < 0.01, ***p < 0.001
The security analysis
Biosafety is a good concern in anti-tumor remedy earlier than medical purposes. We first evaluated the hemolysis ratio of crimson cells. The hemolysis ratio of the Fe-PHCN@DOX was lower than 4.1% on the most focus (200 μg mL−1), suggesting their fascinating blood biocompatibility (Extra file 1: Fig. S34). Furthermore, the potential biocompatibility of the Fe-PHCN@DOX towards main organs was investigated with histology evaluation utilizing H&E staining. No apparent tissue harm or inflammatory lesions had been noticed within the main organs from all remedy teams (Extra file 1: Fig. S35), indicating the wonderful biosafety of the Fe-PHCN@DOX. Moreover, no apparent irregular mouse behaviors amongst all remedy teams had been visualized in the course of the remedy course of. For liver toxicity, serum ranges of AST and ALT within the group of Fe-PHCN@DOX weren’t clearly elevated (Extra file 1: Fig. S36A, B). As certainly one of key inflammatory cytokines, IL-1β was not clearly induced after nano-nuclear reactor handled (Extra file 1: Fig. S36C). In the meantime, H&E staining information didn’t present the apparent tissue harm within the main organs (Extra file 1: Fig. S36D). Due to this fact, the above outcomes revealed that the technique of utilizing nano-nuclear-reactors mediated remedy was wonderful biocompatibility and biosafety, suggesting no apparent unintended effects in vivo.