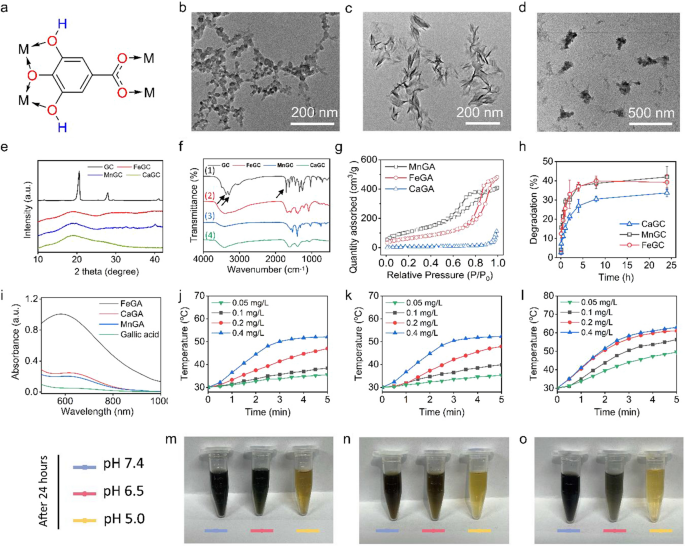
Preparation and characterization of FeGA, CaGA and MnGA metal-gallate nanoparticles
As proven in Fig. 2a, GA has phenolic hydroxyl and carboxyl teams which could be served as natural ligands to covalently bond to metallic atoms. On this examine, metal-gallate nanoparticles had been synthesized by way of a easy and gentle artificial methodology with none nanocarriers, which may keep away from the toxicity and immunogenicity introduced by the nanocarriers [26]. Three various kinds of metallic cations, Ca (II), Fe (III) and Mn (II) had been used for the nanoformulation of gallic acid, and ensuing the formation of three completely different gallic acid-based nanoparticles. As proven in Fig. 2b–d, the as-prepared CaGA, FeGA and MnGA confirmed completely different morphologies and particle sizes. XRD outcomes revealed that CaGA and FeGA nanoparticles had been amorphous, whereas MnGA nanoparticles had been low-crystalline with weak diffraction peaks at 32.6, 34.5 and 40 levels (Fig. 2e). DLS evaluation confirmed the common measurement of FeGA, MnGA and CaGA had been 105.7, 164.2 and 295.3 nm, respectively (Further file 1: Determine S1). Further file 1: Determine S2 illustrates that every kind of metal-gallate nanoparticles had a detrimental zeta potential because of the existence of carboxyl and hydroxyl teams (− 2.9 for FeGA, − 8.1 for MnGA and − 4.5 for CaGA, respectively). FTIR spectra revealed that after the coordination with metallic ions, the stretching vibration of phenolic group at 3200–3400 cm−1 and carboxyl group at 1684 cm−1 (indicated by black arrows) disappeared, indicating that H of GA was changed by metallic ions (Fig. 2f) [27, 28]. As proven in Fig. 2g, the MnGA had a comparatively increased particular floor space (374.8 m2/g). Moreover, the existence of metallic ingredient in every metal-gallate nanoparticles was additional validated by way of EDS as depicted in Further file 1: Determine S3. The degradation charges of metal-gallate nanoparticles in PBS of pH at 7.4 had been round 35–40% (Fig. 2h), which had been ascribed to the breakdown of the reversible coordinate bonds between GA and metallic ions within the presence of hydrogen ion. As proven in Further file 1: Determine S4, the metal-gallate nanoparticles step by step degraded with the pH decrement, and the thorough degradation was achieved when pH reached 5.0, which confirmed the attribute of acid-activated degradation. Further file 1: Determine S5 confirmed that as-prepared metal-gallate nanoparticles exhibited excessive dispersity in deionized water and shaped darkish inexperienced or darkish violet options, revealing the absorption of seen gentle and potential photothermal efficiency of these supplies. As proved by UV–Vis-NIR spectra (Fig. 2i), there was an absorption peak at 580 nm for FeGA, and 615 nm for CaGA and MnGA. As proven in Further file 1: Determine S6, the mass extinction coefficient of the NPs at 808 nm calculated from the Lambert–Beer regulation was 4.0 L g−1 cm−1 (FeGA), 3.25 L g−1 cm−1 (CaGA) and three.01 L g−1 cm−1 (MnGA) respectively. Additional, the photothermal-conversion effectivity (η) of the metal-gallate nanoparticles was calculated in accordance with the earlier method [29]. Primarily based on Further file 1: Determine S7, the η of FeGA, MnGA and CaGA had been 28.2%, 20.4% and 18.9%, respectively. Then the photothermal efficiency of metal-gallate nanoparticles was evaluated by irradiation with a NIR laser of 808 nm and an influence density of two W cm−2. As proven in Fig. 2j–l in addition to Further file 1: Determine S8, FeGA exhibited elevated photothermal conversion effectivity than CaGA and MnGA on account of its comparatively stronger absorption of 808 nm gentle. pH-triggered degradations of metal-gallate nanoparticles had been proven in Fig. 2m–o, that the colour of the answer turned lighter because the pH decreased because of the reversible coordinate bonds between GA and metallic ions within the presence of huge H+. As well as, measurement and NIR absorption are proved to be comparatively secure inside 24 h in DMEM, which ensures the steadiness of nanoparticles in vivo to carry out PTT (Further file 1: Determine S9 and S10). Though Fe ions and gallic acid have been collectively used to assemble therapeutic nanoparticles for anti-cancer functions, the synthesis of those nanoparticles generally includes the usage of carriers, like polyvinylpyrrolidone (PVP), BSA and mesoporous silica [30,31,32]. The multicomponent design inevitably led to unwanted side effects and immunogenicity.
Characterizations of metal-gallate nanoparticles. a Schematic construction of coordination between gallic acid and metallic ions (M characterize metallic ions). b–d TEM photographs of CaGA, MnGA and FeGA nanoparticles. e XRD patterns of FeGA, MnGA and CaGA nanoparticles. f FTIR curves of gallic acid, FeGA, MnGA and CaGA nanoparticles. g Particular floor space of FeGA, MnGA and CaGA nanoparticles. h Degradations of FeGA, MnGA and CaGA nanoparticles in PBS at pH 7.4. i Vis–NIR spectra of GA, FeGA, MnGA and CaGA nanoparticles. j–l Temperature modifications of CaGA, MnGA and FeGA nanoparticles at varied concentrations (0.05, 0.1, 0.2, 0.4 mg/L) after 808 nm NIR laser irradiation. m–o Images of CaGA, MnGA and FeGA in PBS with pH 7.4, 6.5 and 5.0
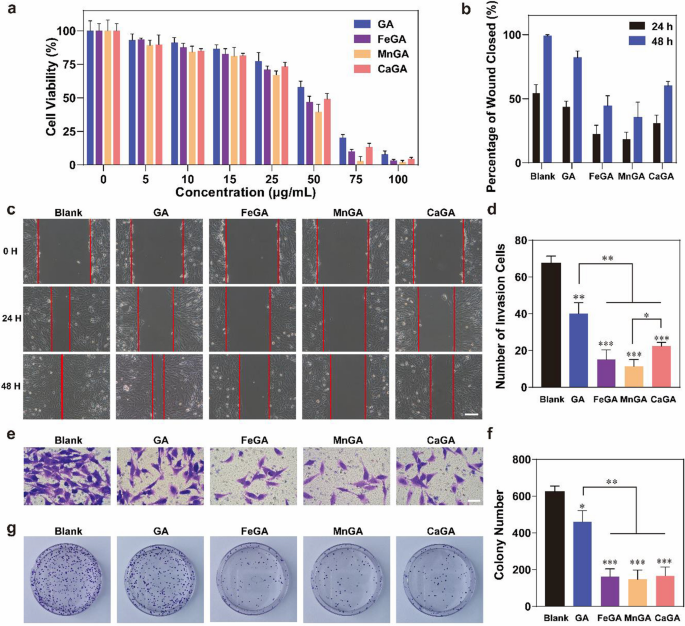
In vitro analysis of anticancer of GA and metal-gallate nanoparticles
To find out the cytotoxicity of GA and metal-gallate nanoparticles towards most cancers cells, completely different concentrations of GA and metal-gallate nanoparticles had been utilized to MG-63 cells for twenty-four h, and their viabilities had been evaluated by CCK-8 assays. As proven in Fig. 3a, in contrast with GA, metal-gallate nanoparticles had an clearly enhanced cytotoxicity towards MG-63 cells when the focus exceeded 25 μg/ml because of the organic exercise of certain metallic ions. Among the many three varieties of metal-gallate nanoparticles, MnGA exhibited the best diploma of cytotoxicity, then by FeGA, whereas CaGA confirmed the bottom cytotoxicity towards the MG-63 cell. Fe and Mn are identified to catalyze the Fenton response in most cancers cells and result in extreme ROS manufacturing and organelle harm [33]. It ought to be famous that Mn(II) ions have the next skill to catalyze Fenton response than Fe(III) ions [34, 35], which can clarify the best cytotoxicity of MnGA nanoparticles.
In vitro anticancer analysis of GA and metal-gallate nanoparticles. a The cytotoxic impact of GA and metal-gallate nanoparticles measured by CCK8. b, c Impact of GA and metal-gallate nanoparticles on cell migration which was measured by way of wound therapeutic assays. d, e The impact of GA and metal-gallate nanoparticles on cell invasion as measured by transwell assays. f, g Impact of GA and metal-gallate nanoparticles on cell proliferation measured by the variety of colonies. *p < 0.05, **p < 0.01, ***p < 0.001. The size bar is c 200 μm, e 100 μm
A wound therapeutic assays in addition to transwell assays had been additionally used to check the results of GA and metal-gallate nanoparticles on tumor cell migration and invasion in vitro. As proven in Fig. 3c and e, GA alone confirmed a sure diploma of skill to inhibit tumor migration, nonetheless, nanoformulated GA with metallic ions confirmed a much-enhanced anti-migration impact, which was extra clearly evidenced by the quantitative information (Fig. 3b and d). Among the many formulated GA nanoparticles, MnGA confirmed a greater inhibitory impact towards MG-63 cells in contrast with FeGA and CaGA. As depicted in Fig. 3f and g, the formulated metal-gallate nanoparticles had an clearly enhanced skill to cut back the clone formation in comparison with the free GA and no apparent distinction within the inhibitory results was discovered among the many three metal-gallate nanoparticles. Contemplating the toxicity of all formulations at concentrations increased than 50 μg/mL, 5 μg/mL was chosen for colony formation assays, and 10 μg/mL for wound therapeutic assays and transwell assays respectively. Collectively, the above outcomes reveal that the nanoformulation of GA considerably enhanced the inhibition of proliferation of cells, migration and invasion of MG-63 osteosarcoma cell traces by GA, and level out the prevalence of MnGA over the others. Though the MnGA nanoparticles present promising antitumor capacities, Fe ions have been prevailingly used to assemble gallic acid-based nanoparticles for anticancer functions, in all probability attributable to their superior photothermal properties (Fig. 2I) [31, 36]. To one of the best of our information, no Mn- gallic acid nanomaterials have been up to now reported.
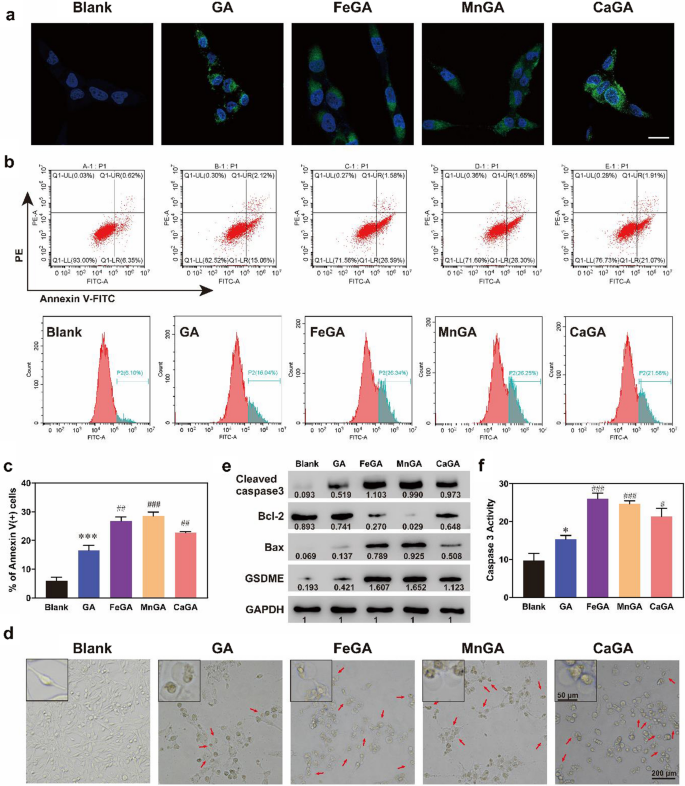
Intracellular ROS technology and pyroptosis induced by metal-gallate nanoparticles
On this examine, intracellular ROS manufacturing skill was detected in MG-63 cells utilizing a ROS probe of two,7-dichlorodihydrofluorescein diacetate (DA-DCFH). Determine 4a confirmed that GA induced slight ROS manufacturing, which was in accordance with earlier research [37]. Nevertheless, FeGA, MnGA and CaGA teams induced extra ROS in cells, as evidenced by the upper fluorescence depth, in comparison with the GA group.
Intracellular ROS technology and pyroptosis induced by metal-gallate nanoparticles. a DA-DCFH fluorescence of MG-63 tumor cells uncovered to PBS, GA, FeGA, MnGA and CaGA Nanoparticles. The size bar is 20 μm. b, c MG-63 cells had been cultured with PBS, GA, FeGA, MnGA and CaGA for six h, and circulation cytometry was used to measure the apoptosis ratios for every group (Annexin V+ cells). d Shiny subject photographs of morphology of MG-63 cells after completely different remedies. e Consultant western blots of N-terminal gasdermin E (N-GSDME), Bax, cleaved caspase-3 and Bcl-2 expressions in MG-63 cells handled with completely different formulations above. f Caspase-3 exercise which was examined by Caspase 3 Exercise Assay Package in MG-63 cells handled with completely different formulations above. *p < 0.05, ***p < 0.001 (when put next with Clean group). #p < 0.05, ##p < 0.01, ###p < 0.001(when put next with group of GA)
It was reported that extreme intracellular ROS may set off mitochondrial permeability transition pore (mPTP) induction and exaggerate ROS manufacturing, which ultimately results in perceptible mitochondrial and mobile damage [38, 39]. Therefore, the mitochondrial dysfunction of MG-63 osteosarcoma cells brought on by metal-gallate nanoparticles analysis was carried out by detecting the modifications of their mitochondrial membrane potential (MMP) by way of 5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazolyl carbocyanine-iodide (JC-1) fluorescent probe. As proven in Further file 1: Determine S11, crimson fluorescence was intense within the PBS group, whereas inexperienced fluorescence was weak, indicating negligible MMP modifications. After remedy with metal-gallate nanoparticles, the cells offered robust inexperienced monomeric fluorescence alerts, representing mitochondrial dysfunctions. To verify if the induced mitochondrial dysfunction may trigger irreversible programmed cell dying, circulation cytometry was used for evaluation of apoptosis. As could be seen from Fig. 4b, c, though GA successfully induced cell apoptosis, the formulated metal-gallate nanoparticles may considerably improve the apoptosis of MG-63 cells. Amongst them, MnGA was superior to others. It’s value noting that the handled MG-63 cells didn’t current a typical morphology of the useless cells by apoptosis. As proven in Fig. 4d, the untreated cells stored uniform elongated morphology, whereas giant quantities of pyroptotic cells had been noticed in GA and metal-gallate nanoparticles group, particularly the metal-gallate nanoparticles group. Massive bubbles blowing from plasma membrane had been discovered within the cells handled with GA or metal-gallate nanoparticles (inset and crimson arrows in Fig. 4d), which is a typical morphological attribute of cell pyroptosis. Not too long ago, it was reported that iron-activated ROS may promote pyroptosis by way of a Bax-caspase3 pathway, and caspase-3 was a pyroptosis inducer by way of cleaving GSDME to GSDME N-terminal [20]. Western blot evaluation of Bax, Bcl2, cleaved caspase-3, and N-terminal GSDME was carried out, and caspase-3 exercise was detected by Caspase 3 Exercise Assay Package. As proven in Fig. 4e, the expression of Bax, a proapoptotic issue with the ability to promote caspase-3, was upregulated by metal-gallate nanoparticles, whereas the expression of Bcl-2, an antiapoptotic protein with the ability to suppress caspase-3 exercise was downregulated. As a consequence, the cleaved caspase-3 and N-terminal GSDME had been upregulated, which is a typical function of pyroptosis. It was obvious that the expression of pro-pyroptotic proteins had been up-regulated by metal-gallate nanoparticles, following an order of MnGA > FeGA > CaGA. Caspase 3 exercise within the cells with metal-gallate nanoparticles remedy was additionally increased than that of GA group, particularly the FeGA and MnGA teams (Fig. 4f). The above outcomes point out that the metal-gallate nanoparticles had been superior to the free GA in enhancing pyroptosis of MG-63 cells, which was possible ascribed to synergistic actions of metallic ions. As proven in Fig. 4a, the metal-gallate nanoparticles enhanced the technology of intracellular ROS. ROS have been reported to activate Bax by way of a Bcl-2-suppressible pathway and thus activate the caspase3 pathway [40, 41]. The canonical caspase-1 inflammasome can induce pyroptosis, or caspases-4, -5, and -11 certain to lipopolysaccharide can straight induce pyroptosis, nonetheless, pyroptosis can also be managed by caspase-3/GSDME as reported in latest research [42]. Zhou et al. [20] reported that Fe ions produced ROS by way of Fenton reactions and activated caspase3-GSDME, which in consequence induced pyroptosis. Subsequently, it’s in all probability that the metal-gallate nanoparticles firstly up-regulated Bax and thus activated caspase-3 by the upregulated intracellular ROS, and in flip GSDME was then cleaved by energetic caspase-3 at its center linker, liberating the gasdermin-N area and forming pores on cell membrane triggering pyroptosis. On this examine, a sure diploma of pyroptosis was discovered within the cells handled with the free GA. Nevertheless, GA-induced pyroptosis has not been reported within the literatures accessible. It was discovered that the concentrations of GA and the remedy period they used had been decrease and shorter, in comparison with these used within the current examine, which presumably accounts for our findings within the completely different position of GA.
Since Ca ions don’t have the power to induce Fenton reactions, nonetheless, the intracellular ROS generated by CaGA was corresponding to these brought on by Fe and Mn ions, and better than that for the GA group. Subsequently, it’s possible indicated that Ca ions synergized with GA in a special unknown method. Tan and Huang et al. [43,44,45] reported that when the capabilities of the endoplasmic reticulum and mitochondria was impaired by medication, the following intracellular enrichment of Ca ions may upregulate the ROS technology. Subsequently, for the CaGA nanoparticle, it’s hypothesized that GA first resulted within the dysfunction of endoplasmic reticulum and mitochondria upon CaGA are uptaken and decomposed, and the launched Ca ions result in the overload of intracellular Ca (II) ion, which finally enhanced ROS technology. The related underlying mechanisms might be additional explored in our future work.
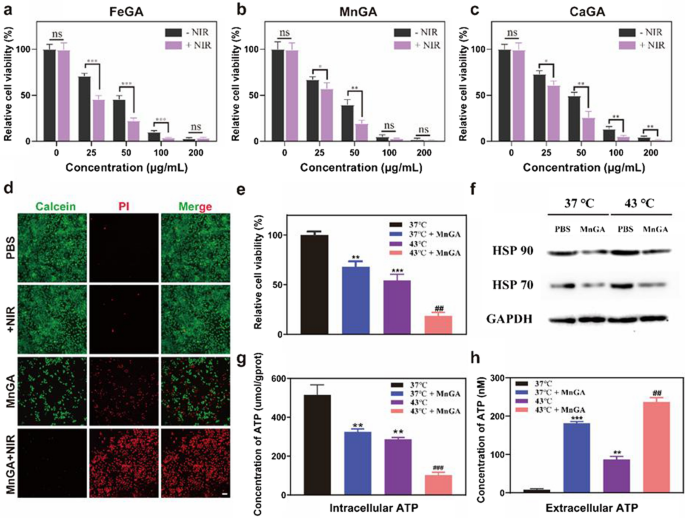
MnGA enhanced mild-temperature PTT by down-regulating HSP by way of exhausting ATP
Provided that tumor cells are much less warmth tolerant than regular cells [46], and hyperthermia may speed up the Fenton response [47], by utilizing PTT and CDT collectively is perceived as a promising technique for enhanced anticancer efficacy. From this facet, the metal-gallate nanoparticles confirmed excessive potential in realizing the combinatory CDT/PTT attributable to their photothermal property and skill to induce intracellular ROS and pyroptosis. To validate this speculation, cytotoxicity experiments had been carried out by treating MG-63 human osteosarcoma cells with metal-gallate nanoparticles of a number of concentrations (0, 25, 50, 75, 100 μg/ml) with or with out NIR irradiation. As compared with the management group, Fig. 5a–c confirmed that metal-gallate nanoparticles teams had a greater skill to destroy tumor cells beneath 808 nm laser irradiation. MnGA nanoparticles had been opted for additional in vitro and in vivo experiments due to their robust anti-cancer skill and relevant photothermal efficiency. Further file 1: Determine S12 confirmed that MnGA might be up-taken by the tumor cells, the quantity of the up-taken nanoparticles elevated because the co-cultivation time was extended. Determine 5d confirmed the pictures of calcein-AM/propidium iodide (PI) stained MG-63 cells handled with PBS and MnGA nanoparticles with and with out NIR irradiation. It may be clearly seen that the MnGA nanoparticle itself induced huge cell dying, indicating its promising chemotherapeutic potential. NIR irradiation (10 min) growing the temperature to 43 °C, nearly killed all of the cells fed with the MnGA nanoparticles, however rendered no hurt to the cells handled with PBS, indicating the potent combinational anticancer results of CDT and PTT of the MnGA nanoparticle.
MnGA nanoparticles improve PTT by down-regulating HSP by way of exhausting ATP. a–c Cell viabilities of MG-63 cells co-incubated with FeGA, MnGA and CaGA nanoparticles for six h at completely different concentrations with and with out irradiation respectively (1.5 W/cm2, 10 min). d Stay/useless photographs of MG-63 cells (stained by calcein and PI) handled with MnGA nanoparticles and PBS on publicity to NIR irradiation or not. Scale bars: 200 μm. e Relative cell viabilities when coincubated with MnGA nanoparticles (15 μg/ml) or PBS for 12 h in 37 or 43 °C. f Intracellular HSP70 and HSP90 protein expression in MG-63 cells in above teams. g Concentrations of intracellular ATP detecting by an ATP Dedication Package. h ATP secretion within the cell medium after varied remedies detecting by an ATP Dedication Package
In conventional PTT, hyperthermia normally with a temperature increased than 50 °C may successfully eradicate tumor cells whereas concurrently leaving damages to regular tissues [48]. Therefore gentle temperature (≤ 45 °C) PTT was proposed to be a promising anti-tumor remedy attributable to its biosafety and biocompatibility [49]. The above outcomes identified the opportunity of MnGA within the mild-temperature thermal remedy (43 °C). To additional validate that the improved cytotoxicity was brought on by the warmth generated from NIR irradiation on the MnGA, MG-63 cells handled with PBS and MnGA had been incubated at 37 °C and 43 °C for 8 h. As could be seen from Fig. 5e, considerably enhanced discount in cell viability was discovered for the group of MnGA + 43 °C, indicating the contribution of warmth to the anti-cancer impact. Earlier examine reported that harm of cell like apoptosis brought on by moderate-temperature thermal remedy (e.g., 45 °C) could be prevented or weakened by the repairing mechanism of cells by way of overexpression of warmth shock proteins (HSP) [50]. To additional perceive the underlying synergistic anti-tumor mechanism of the MnGA, the 2 primary HSP proteins (HSP70 and HSP90) expressions had been studied. HSPs expression has been reported to be depending on ATP consumption [51], therefore mitochondrial dysfunction-induced ATP underproduction can down-regulate the expression of HSP. Moreover, ATP was reported to have the ability to be launched to extracellular matrix (ECM) by way of the pores on pyroptosis cell, and now we have proved that MnGA induced pyroptosis by way of caspase-3-GSDME pathway. Subsequently, it’s of nice necessity to shed gentle into how ATP manufacturing was regulated by MnGA and incubation at evaluated temperatures. As proven in Fig. 5g, each 43 °C incubation and MnGA remedy suppressed the intracellular ATP technology. Moreover, MnGA additionally induced huge ATP efflux into extracellular matrix (Fig. 5h). Subsequently, we consider that the inhibition of ATP technology and the ATP efflux synergistically contributed to the downregulation of HSPs, ultimately enhanced the mild-temperature PTT of the MnGA. Collectively, above outcomes indicated that by exhausting intracellular ATP by way of downregulation of HSPs, MnGA might be an efficient adjuvant for mild-temperature PTT.
In vivo photothermal picture and anticancer analysis
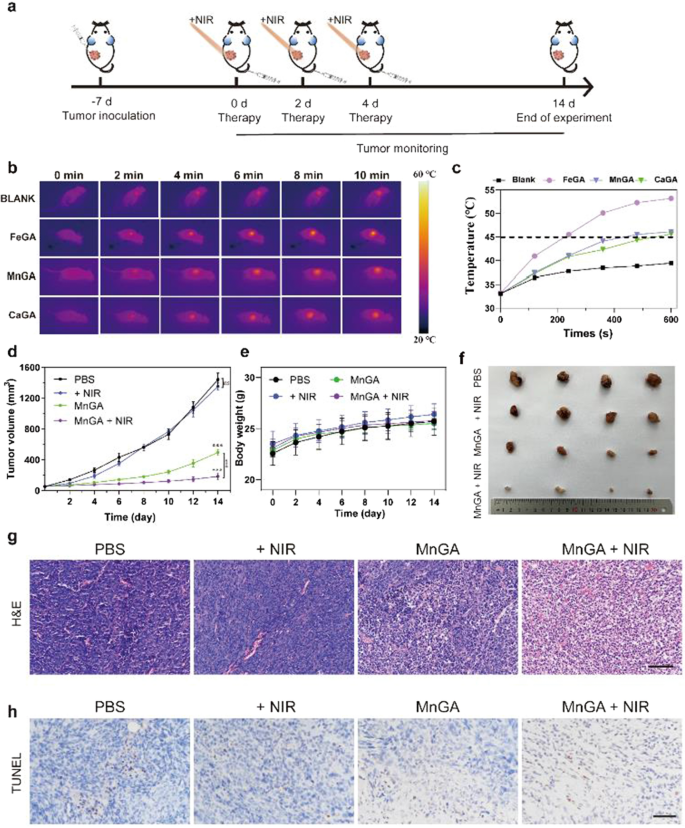
As proven in Fig. 6a, MG-63 osteosarcoma cells had been used to create a mouse subcutaneous tumor mannequin. Briefly, the MG-63 tumor-bearing mice having tumors quantity of about 100 mm3 had been grouped and handled with PBS, + NIR, metal-gallate nanoparticles, metal-gallate nanoparticles plus NIR laser (1.5 W/cm2, 10 min). The NIR irradiation was carried out each two days (day 0, 2 and 4). Earlier than the systematic animal experiment, the in vivo photothermal impact of FeGA, MnGA and CaGA had been assessed utilizing a thermal imager with completely different irradiation period. As proven in Fig. 6b and Fig. 6c, the temperature rose to 54 °C for the FeGA group after NIR irradiation for 10 min, whereas the temperature reached solely to 45 °C for MnGA and CaGA. As we talked about earlier than, excessive temperature ablation of tumors results in a number of heating harm of regular tissues across the tumor [13, 52]. Subsequently, the irradiation period (10 min) comparable to the temperature of 43 °C was chosen for the animal research. Though the photothermal conversion effectivity of FeGA was the best, the MnGA was superior and chosen for the animal research attributable to its potent chemotherapeutic results.
In vivo photothermal photographs and antitumor impact analysis of MnGA-based mild-temperature PTT. a Schematic diagram of the in-vivo experiment to guage the anti-tumor impact of MnGA nanoparticles. b The consultant thermal photographs of the mice with intravenous administration of PBS, FeGA, MnGA and CaGA (10 mg/kg) beneath NIR irradiation for 10 min (1.5 W/cm2). c The corresponding temperature modifications over time curve of tumor areas with injection of PBS, FeGA, MnGA and CaGA. d Tumor quantity progress curves of every group (n = 4). e The physique weight modifications within the 4 teams of mice. f The pictures of the tumors on day 14 after part. g H&E staining of tumor sections in 4 completely different teams. h TUNEL evaluation of the tumor sections from varied teams on the 14th day of the experiments. Scale bars: g, h) 100 μm. ns: no significance, ***p < 0.001
Determine 6d exhibits the tumor progress curves from completely different teams. It may be seen that the NIR group confirmed negligible tumor inhibition, whereas tumors had been inhibited to some extent by the MnGA group. Vital tumor suppression was noticed within the MnGA + NIR group. The physique weight of the mouse for the MnGA and MnGA + NIR teams didn’t present vital distinction from the management group (Fig. 6e), indicating the great biocompatibility of MnGA nanoparticles. The pictures of the first tumors and their weights had been displayed in Fig. 6f and Further file 1: Determine S13, exhibiting an analogous pattern to that noticed in Fig. 6d. Staining with hematoxylin and eosin (H&E) outcomes confirmed that mixed remedy considerably lowered tumor cell harm, characterised by shrinkage and nuclear condensation of the tumor cells (Fig. 6g). Terminal deoxynucleotidyl transferase dUTP nick finish labeling (TUNEL) assay illustrated that the MnGA + NIR group confirmed the best mortality charge of tumor cells (Fig. 6h). For the reason that in vitro experiments proved that MnGA promoted cell dying by induction of pyroptosis apart from apoptosis, and the improved PTT was additionally associated to ATP exhaustion by way of pyroptosis, we additional confirmed that the activation of pyroptosis by MnGA was by way of Caspase-3-GSDME pathway in vivo. As seen in Further file 1: Determine S14, caspase-3 and GSDME-N had been up-regulated in MnGA and MnGA plus NIR group, particularly within the latter group. In abstract, the animal research proved that the MnGA with the NIR irradiation may considerably eradicate tumor ascribed to the mix of the potent chemotherapy and mild-temperature PTT.
In vivo biodistribution and biosafety analysis of MnGA
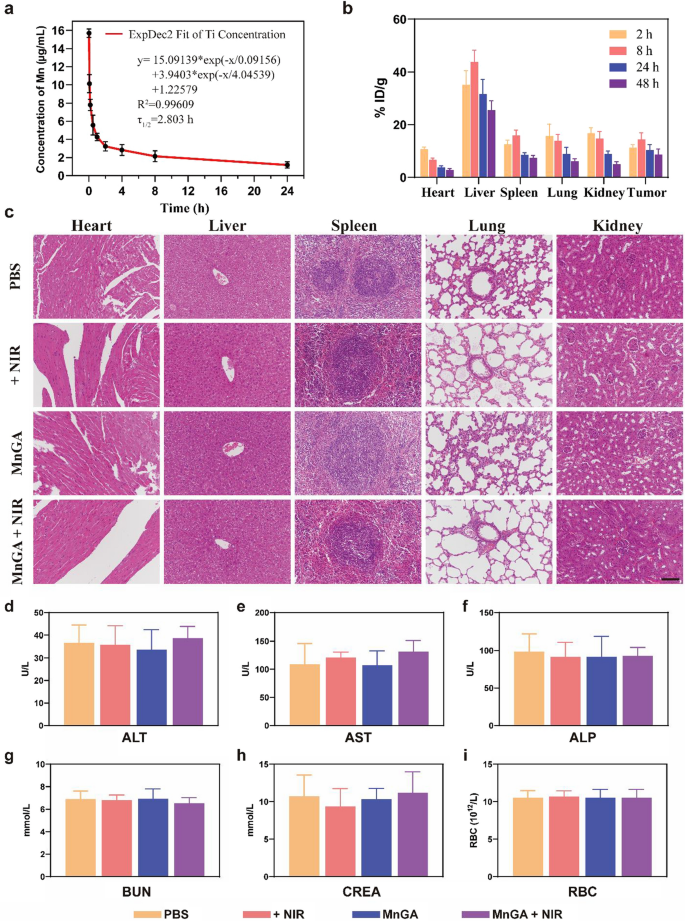
As proven in Fig. 7, in vivo biodistribution and biosafety had been assessed to check toxicity or unwanted side effects induced by MnGA or PTT remedy. Firstly, by utilizing inductively coupled plasma mass spectrometry (ICP-MS), the pharmacokinetics of MnGA had been evaluated in vivo. Observing the blood-circulation curves of Mn ingredient, it may be seen that the MnGA was faraway from blood vessels step by step, and the half-life intervals (τ1/2) in vivo had been about 2.8 h (Fig. 7a). On the idea of the standard EPR impact in stable tumors, MnGA was enriched at tumor websites with a concentrating on effectivity of 14.39% ID/g at 8 h (Fig. 7b).
In vivo biodistribution and biosafety analysis of MnGA Nanoparticles. a Blood-circulation curves of MnGA nanoparticles administrated intravenously (10 mg/kg, n = 4). The half-life interval (τ1/2) of Mn was calculated to approximate 2.803 h. b Biodistributions of MnGA nanoparticles (%ID/g of Mn) in tumors, coronary heart, liver, spleen, lung and kidney after intravenous administration (2, 8, 24 and 48 h, n = 4) c H&E staining of primary organs comparable to the guts, liver, spleen, lung and kidney for biosafety analysis of 4 teams in vivo. d–i Blood biochemical profile and blood routine assessments on the 14th day in teams together with Clean, NIR, MnGA and MnGA + NIR. Scale bar: c 100 μm
Histological evaluation was carried out on the finish of the remedy. As could be seen from the H&E sections (Fig. 7c), primary organs together with coronary heart, liver, spleen, lung, and kidney didn’t present any apparent tissue harm. Moreover, the blood was additionally examined for blood biochemistry and routine assessments. Determine 7d–f confirmed that there was no vital improve in glutamic-pyruvic transaminase (ALT), glutamic oxalacetic transaminase (AST) and alkaline phosphatase (ALP). Likewise, the blood urea nitrogen (BUN) and creatinine (CREA) ranges had been within the regular vary (Fig. 7g and h), indicating no vital harm to the kidneys. In every group, the focus of primary blood cells comparable to crimson blood cells (RBC) was not considerably distinction from that of the management group(Fig. 7i). Usually, these outcomes all collectively proved the biosafety of the developed MnGA nanoparticles, thus, they’ve the potential to be clinically helpful sooner or later.