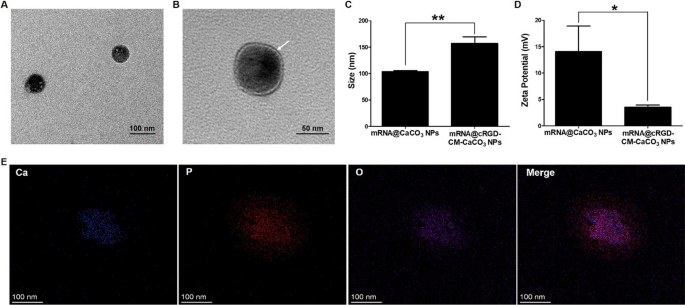
The preparation and characterization of nanoparticles
Firstly, CaCO3 NPs loaded with mRNA (mRNA@CaCO3 NPs) have been ready by means of a reverse microemulsion methodology. Transmission electron microscopy (TEM) noticed that CaCO3 NPs have been spherical in form with a measurement of about 60 nm (Fig. 1A). Then, cell membrane (CM) was derived from GL261 cells by means of repeated freeze–thaw course of. To organize cRGD-labeled CM (cRGD-CM), GL261 cells have been pre-treated with N-azidoacetylmannosamine-tetraacylated (Ac4ManNAz) to connect azide group on the cell floor[25]. After that, for the preparation of CM coated CaCO3 NPs (mRNA@CM-CaCO3 NPs), CM and mRNA@CaCO3 NPs have been blended and co-extruded by a 200 nm polycarbonate membrane. Lastly, click on response was used to switch cRGD on the floor of mRNA@CM-CaCO3 NPs, which was produced between the azide teams of cell floor and the alkyne teams of the pre-synthesized endo-bicyclo[6.1.0]nonyne(BCN)-cRGD (endo-BCN-cRGD). The profitable manufacturing of endo-BCN-cRGD was verified by means of mass spectroscopy (Further file 1: Fig. S1). After cRGD hooked up on the floor of mRNA@CM-CaCO3 NPs, mRNA@cRGD-CM-CaCO3 NPs have been lastly ready. As proven in Fig. 1B, apparent core–shell construction (a visual shell layer of ≈ 8 nm) was noticed in mRNA@cRGD-CM-CaCO3 NPs, indicating profitable CM fusion. The CM coating was additional verified by means of the dimensions and zeta potential modifications detected by means of dynamic gentle scattering (DLS). A rise of common hydrodynamic diameters from 104 nm (mRNA@CaCO3 NPs) to 157 nm (mRNA@cRGD-CM-CaCO3 NPs) was noticed (Fig. 1C). The bigger sizes measured by means of DLS than TEM might attribute to the floor hydration of NPs in DLS detections. The zeta potential was decreased from 14.1 mV to three.5 mV after CM coated (Fig. 1D). In the meantime, the CM coating and mRNA encapsulation will be additionally proved by elemental mapping (Fig. 1E), the place the P ingredient, as a consultant ingredient of CM and mRNA, was properly distributed each inside and out of doors the Ca ingredient. As well as, the encapsulation effectivity of mRNA in mRNA@cRGD-CM-CaCO3 NPs was roughly 70% on the loading capability of practically 2% (mRNA weight/mRNA@cRGD-CM-CaCO3 NPs weight). These outcomes proved that CM was efficiently coated within the floor of mRNA@CaCO3 NPs.
Preparation and characterization of mRNA@cRGD-CM-CaCO3 NPs. A TEM picture of mRNA@CaCO3 NPs cores. B TEM picture of mRNA@CaCO3 NPs cores coated with cRGD-CM shells (mRNA@cRGD-CM-CaCO3 NPs). Arrows present the shells surrounding the cores. C, D Measurement and zeta potential measurement of mRNA@CaCO3 NPs and mRNA@cRGD-CM-CaCO3 NPs. (E) Vitality-dispersive X-ray spectroscopy (EDS) elemental mapping of mRNA@cRGD-CM-CaCO3 NPs. Knowledge are expressed as imply ± SEM (n = 5). *P < 0.05, **P < 0.01
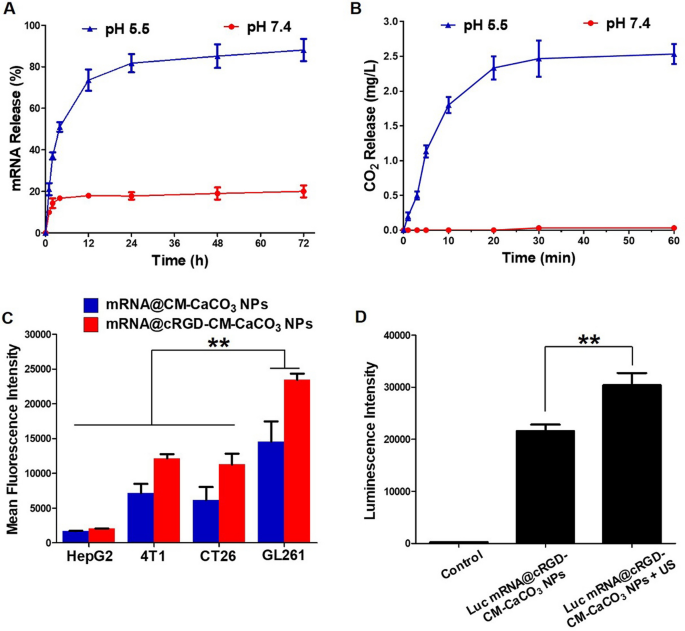
To verify that mRNA@cRGD-CM-CaCO3 NPs might arrive within the tumor web site earlier than decomposition, the pH-dependent launch experiment of mRNA from nanoparticles was carried out. As proven in Fig. 2A, much less quantity of Cy3-labelled mRNA (Cy3-mRNA) was launched from mRNA@cRGD-CM-CaCO3 NPs at impartial situations, demonstrating that CaCO3 NPs have been secure within the systemic circulation. Against this, at pH 5.5 situation, sooner launch of mRNA was noticed, and practically 90% of mRNA was launched after 72 h. These outcomes demonstrated the pH-activated decomposition of CaCO3 NPs. The quantitative evaluation of CO2 fuel era was additional evaluated (Fig. 2B), nearly no CO2 fuel was produced from mRNA@cRGD-CM-CaCO3 NPs at impartial situations. In distinction, a substantial quantity of CO2 was generated at pH 5.5 situation.
A Cumulative mRNA launch from mRNA@cRGD-CM-CaCO3 NPs. B The quantification of CO2 era from mRNA@cRGD-CM-CaCO3 NPs. C Homotypic focusing on by means of fluorescence measurement of nanoparticles incubated in several cell strains. D Luminescence depth of nanoparticles. Knowledge are expressed as imply ± SEM (n = 5). **P < 0.01
Mobile uptake and transfection of mRNA@cRGD-CM-CaCO3 NPs
To check whether or not cRGD-CM-CaCO3 NPs might successfully ship mRNA into mind tumor GL261 cells. Mobile uptake and luciferase transfection assay have been carried out. We first used Cy3-mRNA to detect the mobile uptake effectivity in several cell strains together with HepG2, 4T1, CT26 and GL261 cells by means of stream cytometry (Fig. 2C). When comparability was made amongst all 4 cell strains, the imply fluorescence depth of GL261 cells handled with mRNA@CM-CaCO3 NPs or mRNA@cRGD-CM-CaCO3 NPs have been considerably stronger, suggesting that GL261 CM coating might help CaCO3 NPs enter into GL261 cells by means of the homotypic focusing on impact. Moreover, when comparability was made in every cell line, the imply fluorescence depth was stronger in cRGD-labeled group (mRNA@cRGD-CM-CaCO3 NPs) in contrast with CM coated solely group (mRNA@CM-CaCO3 NPs), which proved that cRGD performed a major position in facilitating the mobile uptake of nanoparticles. Subsequently, we used mRNA encoding luciferase (Luc mRNA) to judge the transfection effectivity of mRNA@cRGD-CM-CaCO3 NPs. In response to Fig. 2D, Luc mRNA@cRGD-CM-CaCO3 NPs have been capable of transfect GL261 cells with a excessive luminescence depth. Moreover, the transfection effectivity of Luc mRNA was additional improved after US irradiation (2776 Mind Cellular Ultrasound System, Chattanooga, USA), which could as a result of enhanced gene supply effectivity by means of US-mediated acoustic cavitation and sonoporation impact [26].
In Vitro immunogenic necroptosis impact of nanoparticles
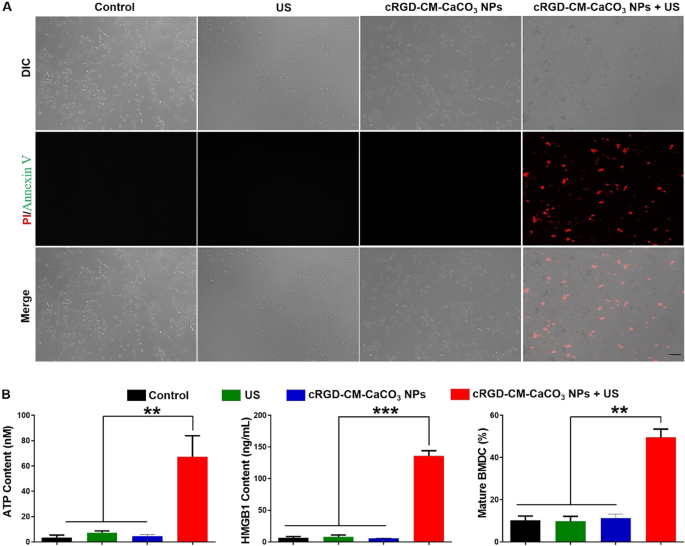
Necroptosis is characterised by membrane rupture and cytoplasmic swelling [27]. To confirm the cell dying mechanism, the annexin V/propidium iodide (PI) assay was carried out in GL261 cells [10, 28]. As illustrated by Fig. 3A, when the cells have been solely handled with US irradiation or cRGD-CM-CaCO3 NPs, no vital modifications within the morphology and no fluorescent indicators from annexin V/PI have been noticed, which proved that US irradiation or cRGD-CM-CaCO3 NPs alone didn’t trigger apparent injury to GL261 cells. Whereas, cRGD-CM-CaCO3 NPs plus US-treated cells displayed a loss of their cell morphology, demonstrating that the cell membrane was broken as a result of US-mediated cavitation impact. Furthermore, the membrane rupture induced the leakage of membrane fragments, cytosolic elements and chromatins [29]. These outcomes validated that the mix of cRGD-CM-CaCO3 NPs and US irradiation might induce necroptosis of GL261 cells. Inspired by the above knowledge, launched DAMPs (together with HMGB1 and ATP) have been additionally evaluated. cRGD-CM-CaCO3 NPs + US group considerably improved the extracellular secretion of HMGB1 and ATP in contrast with different teams. Consequently, the DCs maturation frequency of cRGD-CM-CaCO3 NPs + US group was highest and as much as 49.6% (Fig. 3B).
cRGD-CM-CaCO3 NPs-induced necroptosis and DAMPs launch. A Fluorescence microscope pictures of annexin V (inexperienced fluorescence)/PI (crimson fluorescence)-stained GL261 cells. Scale bar, 100 µm. B Launch of ATP, HMGB1 and bone marrow-derived dendritic cells (BMDCs) maturation after completely different therapies. Knowledge are expressed as imply ± SEM (n = 5). **P < 0.01, ***P < 0.001
Subsequent, cRGD-CM-CaCO3 NPs have been loaded with IL-12 mNRA and the in vitro cytotoxicity was carried out by methyl thiazolyl tetrazolium (MTT) assay. As anticipated (Further file 1: Fig. S2), no vital cytotoxic impact was noticed in cRGD-CM-CaCO3 NPs group. In distinction, cRGD-CM-CaCO3 NPs + US group and IL-12 mRNA@cRGD-CM-CaCO3 NPs group exhibited reasonable cytotoxic impact, which was attributed to acoustic cavitation or the efficacy of IL-12 mRNA respectively. Notably, IL-12 mRNA@cRGD-CM-CaCO3 NPs + US group confirmed the strongest cytotoxicity and killed practically 70% of the cells, implying that the mix remedy of IL-12 mRNA@cRGD-CM-CaCO3 NPs and US irradiation might improve in vitro antitumor impact.
General, these outcomes proved that US-mediated necroptosis leaded to the discharge of DAMPs, which induced the DCs maturation and enhanced antitumor immunity.
In vivo imaging and security analysis
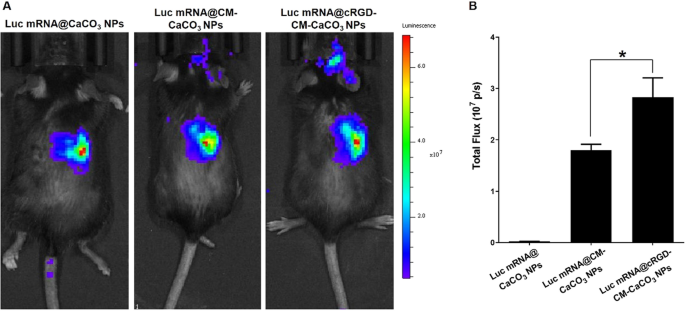
To confirm the mind tumor-targeted of cRGD-CM-CaCO3 NPs in vivo, an intracranial orthotopic glioblastoma (GL261) mice mannequin was used. Luc mRNA@ CaCO3 NPs, Luc mRNA@CM-CaCO3 NPs or Luc mRNA@cRGD-CM-CaCO3 NPs have been intravenously injected at an mRNA dose of 0.25 mg/kg. After 6 h, we measured the bioluminescence indicators by a IVIS imaging system (Fig. 4A). A lot of the Luc mRNA@ CaCO3 NPs collected within the liver, as soon as coated with CM, a part of the Luc mRNA@CM-CaCO3 NPs have been discovered within the mind tumor web site. Extra importantly, after cRGD embellished, Luc mRNA@cRGD-CM-CaCO3 NPs displayed practically 1.6-fold greater bioluminescence sign depth than CM coated alone group (Luc mRNA@CM-CaCO3 NPs) within the glioma space (Fig. 4B). These outcomes demonstrated that CM coated contribute to mind tumor focusing on, and the cRGD modification can additional improve the focusing on functionality.
Subsequent, we evaluated the toxicity of the nanoparticles in wholesome C57BL/6 mice. The measurement of blood biochemistry parameters and HE staining of main organs have been carried out after handled with PBS, IL-12 mRNA@CaCO3 NPs, IL-12 mRNA@CaCO3 NPs + US, IL-12 mRNA@cRGD-CM-CaCO3 NPs or IL-12 mRNA@cRGD-CM-CaCO3 NPs + US. Blood urea nitrogen (BUN) is usually used for assessing renal perform [30]. Aspartate aminotransferase (AST) and alanine aminotransferase (ALT) are efficient predictors of liver pathology [31]. As proven in Further file 1: Fig. S3, there was no apparent variations in BUN, AST and ALT ranges amongst all of the teams, proving that no vital renal and liver toxicity after nanoparticles treating. Furthermore, no obvious histopathological modifications have been discovered within the main organs by means of HE staining (Further file 1: Fig. S4). For additional verifying the protection of US irradiation to the mind, HE staining of mind was carried out after handled with PBS or US irradiation in wholesome C57BL/6 mice. And no obvious histopathological modifications in mind have been discovered after US irradiation (Further file 1: Fig. S5), which proving that therapeutic US irradiation was secure for the conventional mind tissues. All these outcomes demonstrated that CaCO3 NPs plus US irradiation can function a secure technique for tumor remedy.
In vivo anti-glioma exercise
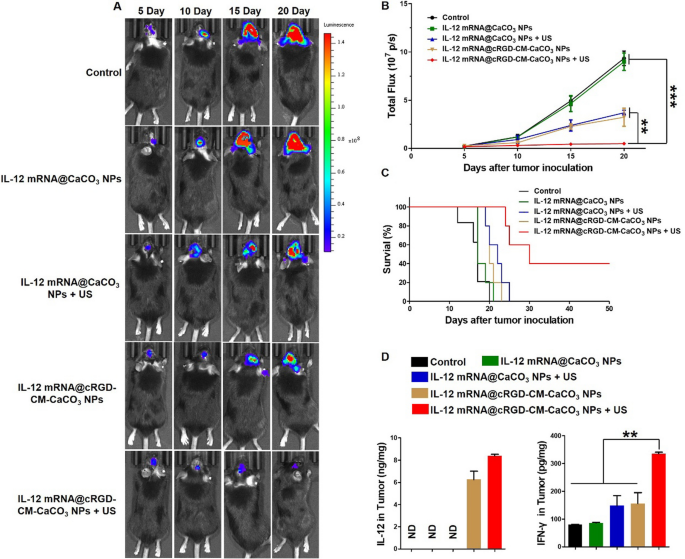
Inspired by the wonderful antitumor results in vitro and brain-targeting capacity in vivo of cRGD-CM-CaCO3 NPs, we investigated antitumor efficacy of the nanoparticles in vivo by an orthotopic GL261-Luc glioma mouse mannequin. As illustrated by Fig. 5A, B, IVIS Spectrum confirmed that speedy tumor development within the PBS or IL-12 mRNA@CaCO3 NPs handled group. Whereas reasonably restricted most cancers development was noticed in IL-12 mRNA@CaCO3 NPs + US group and IL-12 mRNA@cRGD-CM-CaCO3 NPs group. Moreover, the bioluminescence indicators of IL-12 mRNA@cRGD-CM-CaCO3 NPs + US group was clearly weaker than another group, indicating the strongest antitumor impact. As well as, survival examine additionally proved that the mix of IL-12 mRNA@cRGD-CM-CaCO3 NPs and US irradiation can lengthen mice survival and result in a 40% sturdy treatment charge (Fig. 5C). The physique weight of mice was enormously affected by completely different therapies, which is analogous to the development of survival charge (Further file 1: Fig. S6).
In vivo anti-glioma exercise of IL-12 mRNA@cRGD-CM-CaCO3 NPs. A Consultant bioluminescence pictures of GL261-Luc glioma-bearing mice in several teams. B Quantitative evaluation of bioluminescence sign depth. C Survival curve for the mice (n = 5 mice per group). D IL-12 and IFN-γ expression in tumors from immunized mice. Knowledge are expressed as imply ± SEM (n = 5). ND, not detected. **P < 0.01, ***P < 0.001
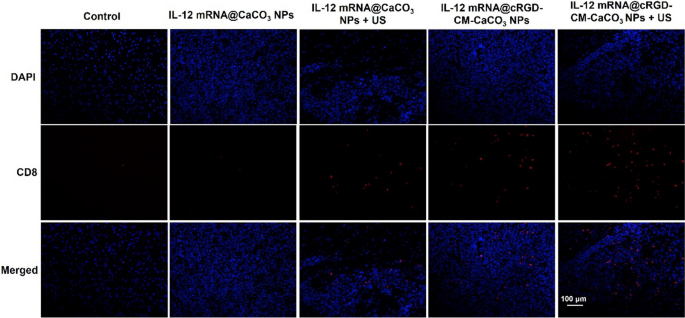
Importantly, IL-12 mRNA@cRGD-CM-CaCO3 NPs remedy plus US considerably elevated the expression of IL-12 in mind tumor sections, in addition to the IFN-γ manufacturing, which is induced by IL-12 instantly (Fig. 5D) [32]. Furthermore, IL-12 mRNA@cRGD-CM-CaCO3 NPs + US group had the most important proportion of CD8 + T cells in tumors in contrast with different teams (Fig. 6). Altogether, these outcomes indicated that the anti-glioma immune response by IL-12 mRNA@cRGD-CM-CaCO3 NPs might be amplified by means of US-mediated necroptosis.