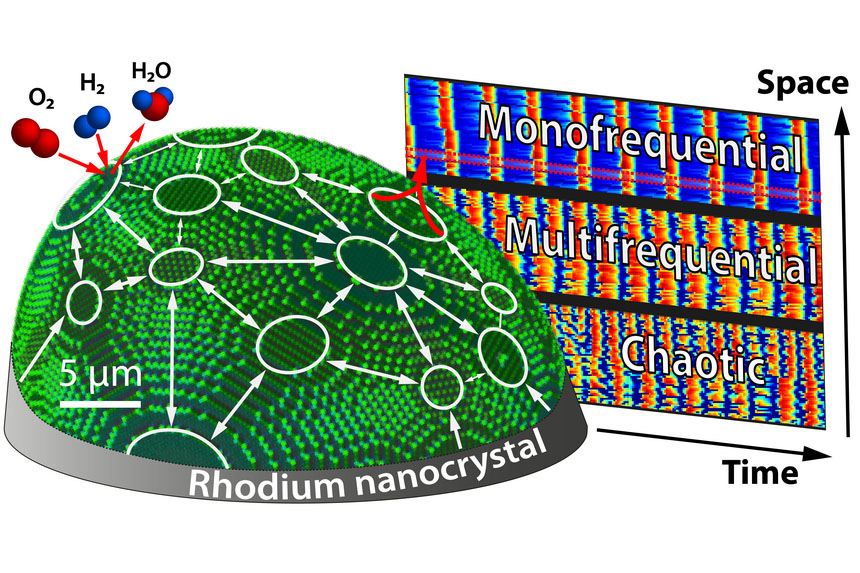
Cell traces
BMDMs had been harvested from mouse femurs to organize mouse macrophages. These cells had been then cultured in Dulbecco’s Modified Eagle’s Medium (DMEM; containing 10% fetal bovine serum (FBS), penicillin/streptomycin, glutamine, and HEPES) and handled with 20 ng ml−1 granulocyte–macrophage colony-stimulating issue (GM-CSF) for 7 days. Subsequent, they had been conditioned with M2 macrophage-conditioned medium (20 ng ml−1 IL-4). Each the murine GL261 and Luci + GL261 cell traces (gifted from Jiangsu College) had been transfected with 10 μg plasmids that encoded cDNAs for ErbB2 (OriGene Applied sciences, RC400096) to acquire the ErbB2 + and Luci + GL261 ErbB2 cell traces (named GL261-H and Luci + GL261-H cell traces), and the Luci + GL261-H cell line was used for in vivo bioluminescent imaging. The RAW264.7 cell line and the 293 T cell line (American Kind Tradition Assortment) had been cultured in full DMEM with 10% heat-inactivated FBS, 100 U ml–1 penicillin and 100 μg ml–1 streptomycin. Human hematopoietic stem cells had been kindly donated by the YINFENG Organic Group, LTD.
Animals
C57BL/6 mice (4 w, ♀) had been bought from Huafukang Biotech. All animal experiments had been accredited by and carried out in compliance with the rules and laws of the Institutional Animal Care and Use Committee of the Cheeloo School of Medication, Shandong College (21074). Animals had been housed in a managed setting, with the next goal situations: temperature, 19–25 °C; relative humidity, 30–60%. An digital time-controlled lighting system was used to supply a 12 h gentle/12 h darkish cycle.
Plasmid era
On this examine, piggyBac transposon gene expression vectors had been used, which contained the CD8α hinge and transmembrane domains and the CD3ζ endo area. The ErbB2-specific scFv area used to generate ErbB2-CAR. This CAR (named CD68 plasmid) assemble with reporter proteins was generated by fusing GFP with the ErbB2-specific scFv area, which was custom-cloned by TsingKe and separated by a T2A sequence. The one distinction between this and the CMV plasmid was using the CMV promoter as a substitute of the CD68 promoter.
C32-122–NLS peptide conjugation
An answer of 12 mg 4-(maleinimido) phenyl isocyanate (PMPI) in DMSO (20 mg ml–1) was added to 86 mg C32-122 polymer in DMSO (100 mg ml–1). The answer was combined at room temperature (RT) for 3 h. The C32-122-maleimide by-product was added to an answer of 100 mg NLS peptide in 5.3 ml DMSO containing tris(2-carboxyethyl) phosphine hydrochloride (TCEP·HCl; 3 mg ml–1). The answer was combined at RT for 3 h after which filtered via a 7 ok Zeba spin column equilibrated with DMSO. The DMSO was evaporated beneath vacuum in a single day. The C32-122-peptide conjugate was redissolved in DMSO to 100 mg ml–1 C32-122 and saved at − 20 °C.
Peptide synthesis
The NLS sequence (Cys-Arg-DArg-Arg-Lys-NH2) and RP-182 peptide (KFRKAFKRFF) had been {custom} synthesized by AnaSpec Inc. A cysteine was added to the N-terminus of the peptide for linkage to the C32-122 polymer and the PGA.
PGA–RP-182 peptide conjugation
PGA was dissolved in water to attain a focus of 20 mg ml–1 after which sonicated for 10 min in a shower sonicator. An equal quantity of ethyl-N′-(3-dimethylaminopropyl) carbodiimide·HCl in water (4 mg ml–1, 16 equiv.) was added, and the answer was combined at RT for five min. The ensuing activated PGA was added to an answer of RP-182 peptide in PBS at a 4:1 molar ratio and combined at RT for six h. Extra reagents had been eliminated by dialysis (1000 MWCO Slide-A-Lyzer Dialysis Cassette) in opposition to PBS for twenty-four h, adopted by filtration via a 40 ok Zeba spin column. The peptide focus was decided utilizing a NanoDrop 2000 Spectrophotometer (Thermo Scientific).
Nanoparticle preparation
All elements had been diluted in sodium acetate buffer (25 mM, pH 5.2) to the next concentrations: DNA, 0.1 mg ml–1; C32-122 and C32-122–NLS, 3.14 mg ml–1; PGA–peptide, 0.45 mg ml–1 peptide. To organize the particles, C32-122–NLS was added to DNA at a PBAE:DNA mass ratio of 15:1. The combination was vortexed gently for 10 s after which incubated at RT for 10 min. Unconjugated C32-122 was then added to the advanced at a PBAE:DNA mass ratio of 15:1. The combination was vortexed gently for 10 s after which incubated at RT for five min. PGA peptide was then added at a peptide:DNA mass ratio of two.5:1. The combination was vortexed gently for 10 s after which incubated at RT for 10 min.
Nanoparticle characterization
The diameters and zeta potentials of the nanoparticles had been decided by a dynamic gentle scattering (DLS) technique utilizing a Malvern Zetasizer Nano ZS90 (Worcestershire, UK). The morphology of the nanoparticles was examined beneath transmission electron microscopy (JEM-1200EX, JEOL). The diluted pattern answer was dropped onto a copper grid adopted by damaging staining with phosphotungstic acid (2 wt%, pH adjusted to six.5 with sodium hydroxide), dried for 20 min after which noticed beneath TEM.
In vitro mobile uptake of FAM-P/PB/N/R nanocarriers (utilizing FAM-modified plasmid as a substitute of the plasmid used to generate P/PB/N/R nanocarriers) by Completely different Cell Varieties.
The mobile uptake of the P/PB/N/R nanocarriers by completely different cell varieties (BMDMs and the RAW264.7, 293 T and GL261-H cell traces) was analyzed. The FAM plasmid was encapsulated into the FAM-P/PB/N/R nanocarriers (FAM-P/PB/N/R nanocarriers) to facilitate detection. For move cytometry evaluation, BMDMs or RAW264.7, 293 T and GL261-H cell traces (2 × 105 cells/properly) had been seeded in six-well plates for twenty-four h. Then, the cells had been incubated with FAM-P/PB/N/R nanocarriers at a plasmid dose of fifty nM. After incubation for six h, the cells had been washed thrice with chilly PBS, trypsinized, and picked up for FCM analyses.
For confocal laser scanning microscopy (CLSM) commentary, BMDMs or RAW264.7, 293 T and GL261-H cell traces (5 × 104 cells/properly) had been seeded onto coverslips in 24-well plates for twenty-four h. After incubation for six h, the cells had been handled with the formulations described above, after which the cytoskeletons and nuclei had been counterstained with rhodamine B(crimson) and DAPI (blue), respectively, based on the usual protocols offered by the suppliers. Then, the mobile uptake habits was visualized beneath CLSM (CLSM 710, Carl Zeiss Inc., Jena, Germany).
In vitro transfection of RAW264.7 cells with artificial DNA nanocarriers
The CD68 plasmid, which concurrently expresses EGFP beneath the management of the CD68 promoter, was used to find out the plasmid transfection effectivity of the artificial DNA nanocarriers, and the CMV plasmid with the CMV promoter was used as a management. Briefly, RAW264.7 cells (2 × 105 cells/properly) had been seeded in six-well plates for twenty-four h, and the cells had been then incubated with artificial DNA nanocarriers with completely different structural integrities (together with free plasmid, P/PB nanocarriers containing plasmid DNA and PBAE, P/PB/N nanocarriers containing plasmid DNA and PBAE C32-122-NLS, P/PB/N/R nanocarriers containing plasmid DNA, PBAE C32-122-NLS and PGA–peptide) at plasmid doses of fifty nM. After incubation for six h, the medium was changed with contemporary medium, and the cells had been additional incubated for 48 h. Afterwards, the share of EGFP-positive cells was analyzed by a move cytometry assay (FACS Calibur move cytometer, BD Biosciences, USA), and after additional incubation for 48 h, EGFP expression was analyzed by western blotting. A monoclonal antibody in opposition to EGFP (Abcam, cat. no. ab184601) was used at a dilution of 1:1000, and goat anti-mouse IgG-HRP (1:5000, Abcam, cat. no. ab6789) was used because the secondary antibody. The outcomes had been visualized utilizing an ImageQuant LAS 4000 mini system (GE Healthcare, UK).
In vitro transfection of various cells with P/PB/N/R
The uptake of the P/PB/N/R nanoparticles by completely different cell varieties (BMDMs and the RAW264.7, 293 T and GL261-H cell traces) was analyzed. FAM-labeled DNA was encapsulated into the P/PB/N/R nanoparticles to facilitate detection. For move cytometry evaluation, BMDMs or RAW264.7, 293 T or GL261-H cells (2 × 105 cells/properly) had been seeded in six-well plates for twenty-four h. Then, the cells had been incubated with nanoparticles loaded with FAM-labeled DNA at a plasmid DNA dose of fifty nM. After incubation for six h, the cells had been washed thrice with chilly PBS, trypsinized, and picked up for FCM analyses.
For CAR expression detection, BMDMs or RAW264.7, 293 T or GL261-H cells (2 × 105 cells/properly) had been seeded in six-well plates for 72 h. After incubation with P/PB/N/R nanocarriers loaded with CD68 plasmid or CMV plasmid for six h, the medium was changed with contemporary medium, and the cells had been additional incubated for 48 h. Subsequently, the share of EGFP-positive cells was analyzed by a move cytometry assay (Beckman Coulter GALLIOS Cytometer, CA, US), and EGFP optimistic cells had been named CAR-MФs.
Macrophage phenotype dedication and cell sorting
All move cytometry antibodies had been bought from Biolegend. Cells had been acquired by move cytometry (Beckman Coulter GALLIOS Cytometer, CA, US). For experiments involving fluorescence-activated cell sorting, a FACSMelody cell sorter (BD Biosciences, US) was used to acquire CAR-RAW264.7 cells by way of GFP.
Cytotoxicity
GL261-H cells had been seeded in 96-well plates and grown for 16 h. CAR-MФs and RAW264.7 cells had been then added to the wells at completely different ratios of 10:1, 5:1, 2.5:1 and 1.25:1 for six h. The discharge of LDH into the medium was quantified utilizing a Cytotox96 nonradioactive cytotoxicity equipment (Promega, Madison, WI, USA) following the producer’s directions. The quantity of LDH launched (% cytotoxicity) was calculated based on the formulation:
$${textual content{Cytotoxicity }}left( % proper),, = ,left( {{textual content{OD49}}0{textual content{ for experimental launch}}, – ,{textual content{OD49}}0{textual content{ for spontaneous launch}}} proper)/left( {{textual content{OD49}}0{textual content{ for max launch}}, – ,{textual content{OD49}}0{textual content{ for spontaneous launch}}} proper), occasions ,{1}00.$$
The spontaneous launch was the quantity of LDH launched from the cytoplasm of GL261-H cells, whereas the utmost launch was the quantity of LDH launched by complete lysis of cells within the experimental group. The assay was replicated thrice.
Phagocytosis assay
The cy5-labeled ErbB2 protein was linked with glutathione-coated polystyrene particles (4.0–4.9 µm diameter, Spherotech) after which resuspended in PBS to be used in phagocytosis assay experiments. The functionalized beads had been incubated with macrophages at a ratio of 1:1 for 45 min, after which the cells had been imaged via high-speed confocal microscopy (Andor Dragonfly 200, UK).
Toxicity research
To measure the potential in vivo toxicities of macrophage-targeted DNA nanocarriers, we injected mice intravenously with varied formulations. Animals had been then euthanized with carbon dioxide to retrieve organs, which had been washed with PBS earlier than fixation in 4% paraformaldehyde. The tissues had been processed routinely, and sections had been stained with hematoxylin and eosin.
Immunofluorescence staining
Tumors had been collected from the mice and snap-frozen in optimum chopping temperature medium. Tumor sections had been reduce utilizing a cryotome, mounted on slides and stained with completely different major antibodies, together with CD8 (Abcam, cat. no. ab22378) and CD80 (Abcam, cat. no. ab100790), in a single day at 4 °C following the producer’s directions. Following the addition of fluorescently labeled secondary antibodies (goat anti-rat IgG (H + L; Thermo Fisher Scientific, cat. no. A18866) and goat anti-rabbit IgG (H + L; Thermo Fisher Scientific, Cat. no. A32733)), the slides had been analyzed with high-speed confocal microscopy (Andor Dragonfly 200, UK). All antibodies used within the experiments had been diluted 200 occasions.
Western blotting
Equal quantities of protein (measured utilizing a bicinchoninic acid protein assay equipment, BCA) had been combined with an equal quantity of two × Loading buffer and boiled at 95 °C for five min. After gel electrophoresis and protein transformation, anti-ErbB2 antibody at a dilution of 1:1,000 (Abcam, Cat. no. ab134182) and anti-GAPDH antibody (Abcam, Cat. no. ab9485) at a 1:5,000 dilution had been used as major antibodies. The secondary antibody used for these blots was a goat anti-mouse antibody (Abcam, Cat. no. ab6721).
In vivo tumour fashions and remedy
Briefly, 5 × 105 Luc + GL261-H cells had been resuspended in 8 μl of chilly PBS and stereotactically implanted into the pons of a mouse (1 mm proper to and three mm posterior to lambda, and three.5 mm depth from the cranium). After loading the injection into the animal, the pores and skin was glued to shut the incision. Free plasmid and varied nanoformulations had been intratumourally injected into GL261-H-bearing mice at a plasmid dose of two mg kg−1 physique weight on day 7, and the PBS group underwent the identical approach of injection with equal quantity of PBS concurrently. In the meantime, the tumour tissues had been collected on days 0 and three for move cytometry evaluation on immune cells (T cells, B cells and macrophages) stained with antibodies to confirm the share of FAM-positive cells and EGFP-positive cells, respectively. As well as, the tumour tissues had been additionally collected on days 0, 7, and 28 for move cytometry evaluation to quantify EGFP expression in macrophages. The tumour volumes had been decided by way of in vivo non-invasive optical biophotonic imaging (PerkinElmer IVIS Spectrum) each 7 days. The mice’s physique weights and survival charges had been monitored concurrently. Moreover, on day 14, the tumour tissues had been harvested and glued in formalin and embedded in paraffin, and 10 μm sections had been reduce to disclose coronal views of the brains. The slices had been processed for normal H&E staining after which had been counterstained with DAPI, and the tumour space was calculated through the use of ImageJ software program.
Luc + PDG cells had been injected stereotactically into the best striatum of huHSC-NOG-EXL feminine mice. PBS and P/PB/N/R was administered intratumourally into huHSC-NOG-EXL tumour bearing mice 7 days. The tumour dimension was monitored each week via BLI.
Circulate cytometry
Tumors collected from mice had been divided into small items and homogenized in chilly staining buffer to type single-cell suspensions within the presence of digestive enzyme. Cells had been stained with fluorescence-labeled antibodies in opposition to CD45 (Biolegend, Cat. no. 103116, clone 30-F11), CD3 (Biolegend, Cat. no. 100217, clone 17A2), CD4 (Biolegend, Cat. no. 100429, clone GK1.5), CD8a (Biolegend, Cat. no. 100721, clone 53–6.7), Foxp3 (Biolegend, cat. no. 126404, clone MF-14), NK-1.1 (Biolegend, Cat. no. 108709, clone PK136), CD62 L (Biolegend, Cat. no. 104407, clone MEL-14), CD44 (Biolegend, Cat. no. 103005, clone IM7), CD11b (Biolegend, Cat. no. 101205, clone M1/70), CD206 (Biolegend, Cat. no. 141731,clone C068C2), CD80 (Biolegend, Cat. no. 104733, clone 16-10A1), F4/80 (Biolegend, Cat. no. 123131, clone BM8), Ly-6G (Biolegend, Cat. no. 127615, clone 1A8), Gr-1 (Biolegend, Cat. no. 108407, clone RB6-8C5), CD86 (Biolegend, Cat. no. 105011, clone GL-1), CD103 (Biolegend, Cat. no. 121415, clone 2E7), and CD11c (Biolegend, Cat. no. 117307, clone N418), and had been handled with Fixable Viability Dye (Biolegend, cat. no. 423101), Cell Stimulation Cocktail (Biolegend, cat. no. 423303), Intracellular Staining Perm Wash Buffer (10X) (Biolegend, Cat. no. 421002), and Fixation Buffer (Biolegend, Cat. no. 420801).
MRI
On the endpoints of varied therapies, the mice had been subjected to in vivo T2-weighted MRI research through the use of the next parameters: repetition time (TR)/echo time (TE) = 2500/35 ms; acquisition matrix = 256 × 256; area of view = 30 × 30 mm; variety of slices = 22; slice thickness = 0.4 mm, Fast Imaging with Refocused Echoes (RARE) issue = 8.
Human tumor specimens
Affected person tumor sections had been obtained from the Division of Neurosurgery at Shandong Provincial Hospital Affiliated with Shandong First Medical College. All of the samples had been obtained with written knowledgeable consent and picked up utilizing a normal protocol accredited beneath the Assessment Board of Shandong First Medical College (Approval no. 2020-272). The sections had been incubated with major antibodies in opposition to ErbB2 (dilution of 1:1000; Abcam, cat. no. ab134182).
Statistical evaluation
Every determine legend denotes the statistical take a look at used. Until in any other case indicated, all knowledge are proven because the means ± normal deviations (SD). Statistical evaluation was carried out by one-way evaluation of variance (ANOVA) in GraphPad Prism 7.0 (GraphPad). For all figures, vital variations between teams are indicated by * P < 0.05, ** P < 0.01, *** P < 0.001 and **** P < 0.0001. Outcomes with p values of < 0.05 had been thought-about statistically vital in all analyses (95% confidence stage).