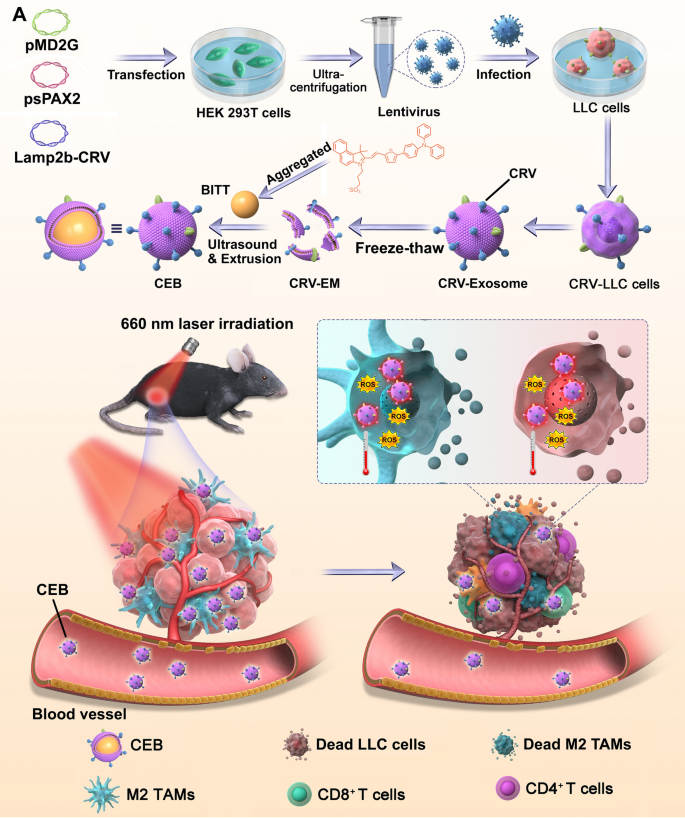
Cell tradition
Lewis lung most cancers cells (LLC), mouse embryonic fibroblast (NIH 3T3), mouse lung epithelial sort II cell (MLE12), mouse embryonic fibroblasts (MEF), and human embryonic kidney epithelial cells (HEK 293T) have been obtained from the American Sort Tradition Assortment (ATCC, USA). The cells have been cultured in DMEM supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin. Cells have been stored in an incubation chamber humidified environment at 37 ℃ and 5% CO2.
Development of CRV-expressed LLC cell line
HEK-293T cells have been plated in 150 mm dishes to 30–50% confluence and altered to a transfection medium, Opti-MEM. A quantity of 25 μL Lipofectamine 2000 was diluted by 50 μL Opti-MEM and incubated for five min. 5 μg pMD2G, 5 μg psPAX2, and 10 μg pcDNA3.1-DNA-Lamp2b-CRV plasmid was combined gently with 50 μL Opti-MEM medium. The plasmid resolution was added to diluted Lipofectamine 2000 and incubated for 20 min. The combination was added to the tradition medium of HEK-293T cells. After the incubation for six h, the cells have been modified to recent medium and cultured for 48 h. The medium was collected and altered to recent media, incubating for one more 24 h to do the second harvest. The collected medium was centrifuged at 3 000 rpm for 15 min at room temperature to pellet cell particles. The virus-containing supernatant was filtered by means of a 0.45 μm and saved at − 80 °C.
LLC cells have been plated in a 6-well plate to 30–50% confluence and altered to Opti-MEM containing virus-containing supernatant. After incubation for six h, the cells have been modified to recent DMEM supplemented with 10% FBS and cultured for 48 h.
Isolation of CRV-exosomes and extraction of exosome membranes
For the isolation of the CRV-Exosomes, the CRV-expressed LLC cells have been cultured in 150 mm dishes for 48 h. The medium was collected and centrifuged at 3000 g for five min at 4 °C to take away cells. The CRV-Exosomes have been obtained by centrifuging supernatant at 10,000 g for 15 min at 4 °C adopted 100,000 g for 70 min at 4 °C and saved at − 80 °C. A N30 move nanoanalyzer (NanoFCM, Fujian, China) was used to review the particle dimension and focus of the exosomes. The morphology of the exosomes was noticed by JEM-2100Plus 200 kV TEM (JEOL, Tokyo, Japan).
To extract the exosomes membranes, the obtained CRV-Exosome precipitation was resuspended in Membrane and Cytosol Protein Extraction Package reagent A (Beyotime Biotechnology, Shanghai, China) containing 1 mM phenylmethylsulfonyl fluoride. After incubation for 15 min in an ice tub, the combination was freeze-thawed for five cycles. The combination was centrifuged at a velocity of 14,000 g for 30 min at 4 °C. The CRV-EM have been quantified with a Pierce BCA Protein Assay Package and saved at − 80 °C. CRV-expressed EM or EM with out CRV have been recognized by Lamp2b, FLAG, ALIX, and HSP70 by means of WB evaluation.
Preparation and characterization of CEB
BITT was gifted from Ben Zhong Tang group in The Hong Kong College of Science and Expertise. Nuclear Magnetic Resonance (NMR) evaluation was carried out to determine the molecular construction of BITT. To organize BITT nanoparticles, BITT was dissolved within the DMSO at a focus of 1.0 mg/mL, which was added to the aqueous resolution at a quantity ratio of 1 to 9 within the presence of water tub ultrasonic vibration at an influence of 600 W for five min. The answer was dialyzed in opposition to deionized water with the dialysis bag (MWCO, 3500 Da) for twenty-four h, which was concentrated by centrifugation at a velocity of 14000 g.
To acquire the CRV-EM coated BITT (CEB), BITT nanoparticles was blended with CRV-EM at totally different ratio (w/w = 1:1, 1:2, 1:5, 1:10, 1:20), adopted by 10 occasions extrusion by means of 200 nm polycarbonate porous membranes. CEB was analyzed with a Nano ZS90 zetasizer (Malvern, Worcestershire, UK).
The freshly ready BITT nanoparticles and CEB as described above, have been added to carbon-coated copper grids, adopted by air drying at room temperature. The evaluation was carried out with a transmission electron microscope. The UV spectrophotometry, excitation, and emission spectra of BITT and CEB have been measured by an UV-1800 spectrophotometer (Shimadzu, Japan) and a RF-6000 fluorospectrophotometer (Shimadzu, Japan).
The protein focus of CRV-LLC cell membranes and CEB have been examined with a Pierce BCA Protein Assay Package. All samples diluted with SDS-PAGE loading buffer have been boiled at 100 ℃ for five min. Afterward, every pattern with an equal protein of 30 μg was loaded onto 10% SDS-PAGE gels and separated by way of gel electrophoresis. The gel was stained with Coomassie blue for 1 h, adopted by washing the gel repeatedly till clear. The information was recorded with an Amersham Imager 600 (GE Healthcare Life Sciences, Little Chalfont, UK).
CEB (BITT equal to fifteen μg/mL) was irradiated by a LR-MFJ-660/1300mW laser (Changchun laser expertise Co., LTD.) for five min at totally different energy to guage its photothermal conversion efficiencies. Temperature modifications have been monitored with a FOTRIC 220 s thermal imaging digital camera (Shanghai InfraRed Techniques Co., LTD, China). CEB (BITT ranged 10–20 μg/mL) was irradiated by 660 nm laser for five min on the depth of two W/cm2. Temperature modifications have been file as above. BITT and CEB (BITT equal to fifteen μg/mL) have been uncovered to a 660 nm laser on the energy of two W/cm2 for five min and eliminated, adopted by repeating 4 occasions. Temperature modifications have been monitored with the thermographic digital camera.
The era of ROS induced by CEB was evaluation. Completely different formulations (BITT equal to fifteen μg/mL) have been incubated with singlet oxygen sensor inexperienced (SOSG) probe (5 μM) and uncovered to 660 nm laser for five min with the facility of two W/cm2. PBS was used as management. The emission spectrums vary have been scanning at Ex = 504 nm with RF-6000 fluorescence spectrophotometer (Shimadzu, Japan).
Mobile uptake of CEB in vitro
LLC cells have been seeded in confocal dishes or 6-well plates (5 × 105 cells per properly). After being cultured in a single day, the cells have been incubated with CEB at totally different doses (BITT equal to five, 10, 15, 20 μg/mL) for six h, respectively. Cells within the confocal dishes have been stained with actin-tracker inexperienced and DAPI for visualization by a Zeiss 880 confocal laser scanning microscope (Zeiss, Germany). Cells in 6-well plates have been analyzed by an ImageStreamX Imaging move cytometer (Merck Millipore, USA) to quantify the quantities of BITT-positive cells after digesting by 0.25% tyrisin and resuspending in PBS.
We additionally evaluated the mobile uptake of CEB over time by CLSM or FACS. Briefly, LLC cells in confocal dishes or 6-well plates have been incubated with CEB (BITT equal to fifteen μg/mL) for various occasions (1, 3, 6, and 12 h). Cells have been stained with actin-tracker inexperienced and DAPI for visualization by CLSM. Intracellular fluorescence depth was quantified by FACS evaluation.
After optimizing the dose and time, totally different formulations, equivalent to BITT, EB, or CEB, have been incubated with LLC cells, respectively. CLSM and FACS evaluation have been utilized to observe the fluorescence.
The mobile uptake of CEB by M2 macrophages was measured. RAW 264.7 cells have been seeded both in confocal dishes or in 6-well plate (2 × 105 cells per properly) and handled with IL4 (50 ng/mL) for 48 h. BITT (focus vary 5–20 μg/mL) was added to the tradition medium, and cells have been cultured for 1, 3, 6, and 12 h. Cells have been noticed upon CLSM after being stained by actin-tracker inexperienced and DAPI or calculated optimistic cells by FACS. The induced M2 macrophages have been incubated with BITT, EB, or CEB based on the optimized situation. The fluorescence was captured by CLSM and FACS.
CEB was ready as described above. CEB (BITT equal to fifteen μg/mL) was incubated with non-specific cell traces, together with NIH 3T3 cells, MLE12 cells, and MEF cells for six h at 37 °C. The cells have been washed 3 times with PBS. For CLSM evaluation, the cells have been fastened and stained with actin-tracker inexperienced and DAPI. To quantitatively analyze mobile uptake, the adherent cells have been indifferent from the tradition plate with 0.25% Trypsin–EDTA Answer and suspended in 100 μL PBS. The cell suspension was analyzed by FACS.
Evaluation of ROS era in cells
The ROS era induced by BITT-based formulations was detected by the reactive oxygen species assay equipment. LLC cells and M2 macrophages cultured in confocal dishes have been handled with PBS, PBS with Laser (PBS + Laser), CEB, BITT with Laser (BITT + Laser), EB with Laser (EB + Laser), and CEB with Laser (CEB + Laser) for six h. DCFH-DA diluted in FBS-free DMEM to 10 μM was incubated with the cells at 37 °C for 30 min at midnight. FBS-free DMEM washed cells to take away DCFH-DA that didn’t enter cells. Teams with laser have been irradiated by a 660 nm laser (2 W/cm2, 5 min). CLSM was utilized to look at the fluorescence at Ex = 488 nm and Em = 525 nm.
Cell viability in vitro
The cells viability of BITT primarily based nanoparticles with or with out laser irradiations was measured by cell counting equipment 8 (CCK-8) assay. LLC cells and polarized M2 macrophages have been seeded in 96-well plate (5 × 103 cells/properly) and handled with totally different formulations (BITT equal to 0 ~ 20 μg/mL) for six h. Cells have been carried out with or with out irradiation by laser (660 nm, 2 W/cm2) for five min. After incubation for 4 h, the medium was discarded, and 10% CCK-8 resolution was added. After incubation for one more 2 h, the absorption at 450 nm was measured. The viability was calculated by the method:
$${textual content{Cell viability }}left( % proper), = ,left[ {left( {{text{A}}_{{{text{sample}}}} – {text{A}}_{{text{blank well}}} } right)/ , left( {{text{A}}_{{{text{control}}}} – {text{A}}_{{text{blank well}}} } right)} right] , *{1}00% .$$
Cell inhibition in vitro
LLC and M2 macrophages have been seeded in confocal dishes and cultured in a single day. PBS, PBS + Laser, CEB, BITT + Laser, EB + Laser, or CEB + Laser was added to the cells. After incubation for six h, the cells have been uncovered to a 660 nm laser for five min on the depth of two W/cm2. After the cells have been cultured for one more 4 h, cells have been stained with Calcein-AM and PI for 15 min, the stay and lifeless cells have been imaged by CLSM.
The analysis of CEB in 3D tumor spheroids
Agarose was dissolved in DMEM to acquire a 2% resolution. The answer (60 μL) was used to coat the underside 96-well plate. LLC cells have been seeded within the agarose-coated plate (4 × 103 cells/properly) and cultured for five days. After forming ~ 500 μm 3D tumor spheroids, the tumor spheroids have been transferred to the agarose-coated confocal dished and handled with BITT, EB, or CEB for twenty-four h. The mobile spheroids in confocal dished have been stained with DAPI for 30 min and imaged by the Z-stack operate of CLSM.
To check the phototherapeutic impact on 3D tumor spheroids, the spheroids have been incubated with totally different formulations (PBS, PBS + Laser, CEB, BITT + Laser, EB + Laser, CEB + Laser) for twenty-four h, and additional uncovered to a 660 nm laser for five min on the depth of two W/cm2. The spheroids have been incubated within the DMEM accomplished medium for one more 4 h. Dwell/Useless Cell Viability Assay Package was used to stain the spheroids and imaged with a CLSM.
Animal experiments
The 4 ~ 6-weeks-old C57BL/6 male mice have been bought from Guangdong Medical Laboratory Animal Middle (Foshan, China) and raised within the particular pathogen-free animal room. We carried out the animal experiments based on the Institutional Authority for Laboratory Animal Care of Guangzhou Medical College (GY2021-142).
Analysis of circulation lifetime in vivo
The C57BL/6 male mice have been administrated with totally different formulations (PBS, BITT, EB, or CEB; n = 3, BITT equal to 200 μg per mouse) by way of intravenous tail vein injections. A quantity of 20 μL of complete blood was collected at time factors 1, 2, 4, 6, 8, 12, and 24 h. The samples have been combined with 0.1 mM acid citrate dextrose and stored at 4 °C till detected by a fluorescence spectrophotometer.
Distribution of CEB in vivo
The C57BL/6 male mice have been subcutaneously inoculated with 3 × 106 LLC cells per mouse on the fitting shoulder to assemble the tumor-bearing mouse mannequin. When tumor volumes reached roughly 100 mm3, the mice have been administered with PBS, BITT, EB, or CEB (BITT equal to 200 μg per mouse) by way of intravenous tail vein injections and monitored with an IVIS Lumina XRMS Sequence III in vivo monitoring system (PerkinElmer, USA). The time factors have been set at 2, 4, 8, 12, 24, and 48 h, respectively. The mice have been sacrificed, and the tumors and main organs have been extracted and imaged. The fluorescence was analyzed with Residing picture software program.
Antitumor impact of CEB in vivo
The xenograft tumor mouse mannequin was constructed by subcutaneously inoculating with 3 × 106 LLC cells per mouse on the fitting flank of C57BL/6 male mice. When the tumor volumes reached roughly 100 mm3, the mice have been randomly divided into six teams (n = 5 per group): PBS, PBS + Laser, CEB, BITT + Laser, EB + Laser, CEB + Laser (BITT equal to 200 μg per mouse). The mice have been administered totally different formulations by way of intravenous tail vein injections each different day and acquired a 660 nm laser irradiation (2 W/cm2, 10 min) 8 h later. The tumor volumes have been measured with a Vernier caliper and calculated as V = (L × W × W)/2. The load of mice was monitored as properly. After the remedy for 13 days, the mice have been sacrificed to gather organs and tumors. Tumors have been organized in orderliness and photographed. The main organs and tumors have been saved in 4% paraformaldehyde and analyzed with Hematoxylin and eosin staining (HE) and TdT-mediated dUTP Nick-Finish Labeling (TUNEL).
The impact of CEB on tumor microenvironment remolding
To find out the in vivo reworking of tumor microenvironment, the quantity of M1 and M2 TAMs, CD8+ T cells, CD4+ T cells, and MDSCs have been decided by FACS and CLSM evaluation. Briefly, tumor-bear mice have been handled with totally different formulations as described above. After the remedies, the mice have been sacrificed, and the tumors have been reduce into items and digested with collagenase IV at 37 °C for 20 min below vibrating. The mixtures have been homogenized with a 40 μM strainer. After the lysis of pink blood cells, cells have been washed with HBSS and divided into 3 tubes. The APC-Cy7-anti-CD45, PE-anti-F4/80+, BV421-anti-CD80, and AF647-anti-CD206 antibodies have been added to the collected cells for 30 min on ice. The staining options have been eliminated and washed with PBS, adopted by fixing the cells with 4% paraformaldehyde. To investigate the T cells, APC-Cy7-anti-CD45, BV510-anti-CD3e, FITC-anti-CD8a, and APC-anti-CD4 antibodies have been added to the cells and incubated on ice for 30 min. MDSCs have been recognized by the addition of APC-Cy7-anti-CD45, PE-anti-Gr1, and Percp-Cy5.5-anti-CD11b antibodies on the ice for 30 min as properly. All of the samples have been analyzed by FACS.
Moreover, the tumors have been reduce into 4-μm-thick sections and examined by immunofluorescence. M1 and M2 TAM have been labeled with anti-CD80 and anti-CD206 antibodies. Anti-CD8 and anti-CD4 antibodies have been utilized to label T cells. MDSCs have been recognized with anti-Ly6G and anti-CD11b antibodies. Curiously, the suppression of vascularization was detected by anti-CD31 and anti-α-SMA antibodies. Immunofluorescence sections have been imaged by CLSM.
Statistical evaluation
The information are expressed because the imply ± commonplace deviation. Information analyses have been carried out utilizing GraphPad Prism 8.0 software program. A two-tailed Pupil’s t check for a two-group comparability was used to research the information. Statistical variations are proven as *p < 0.05, **p < 0.01, and ***p < 0.001.